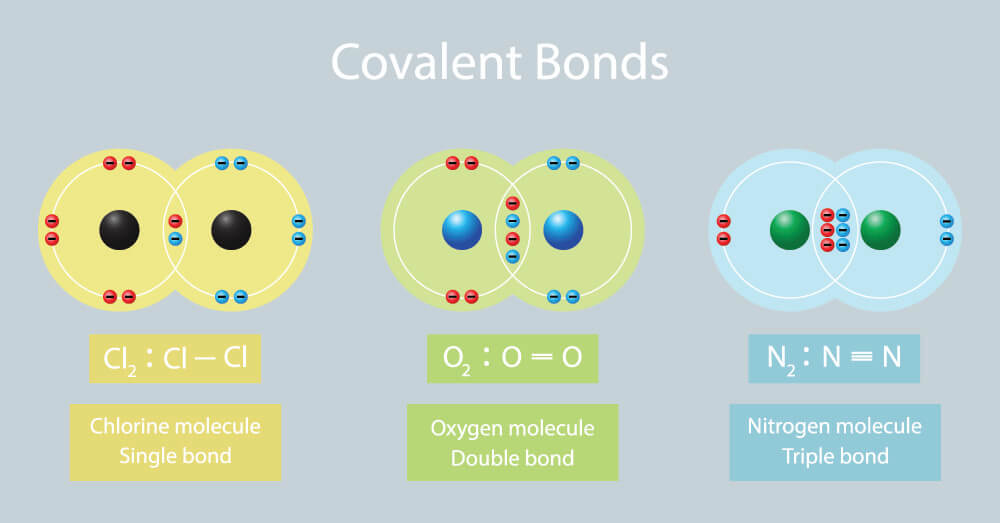
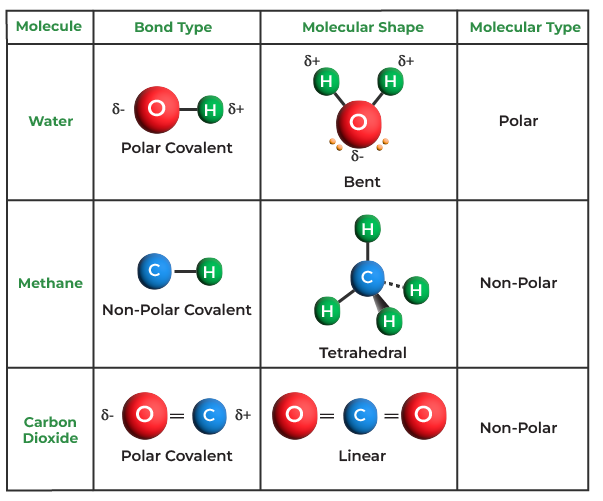
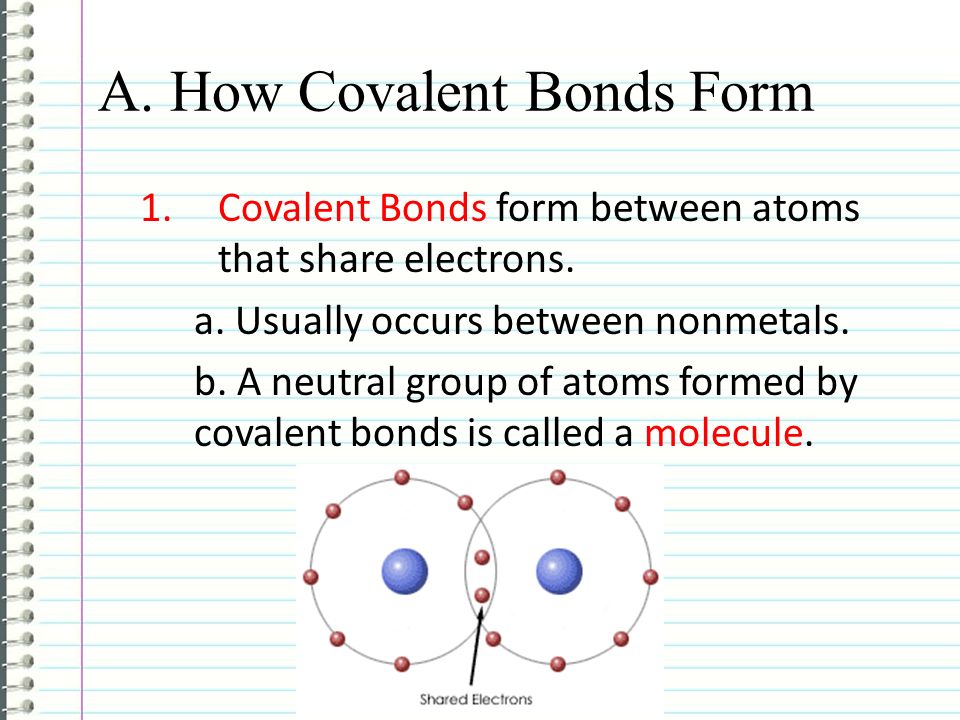
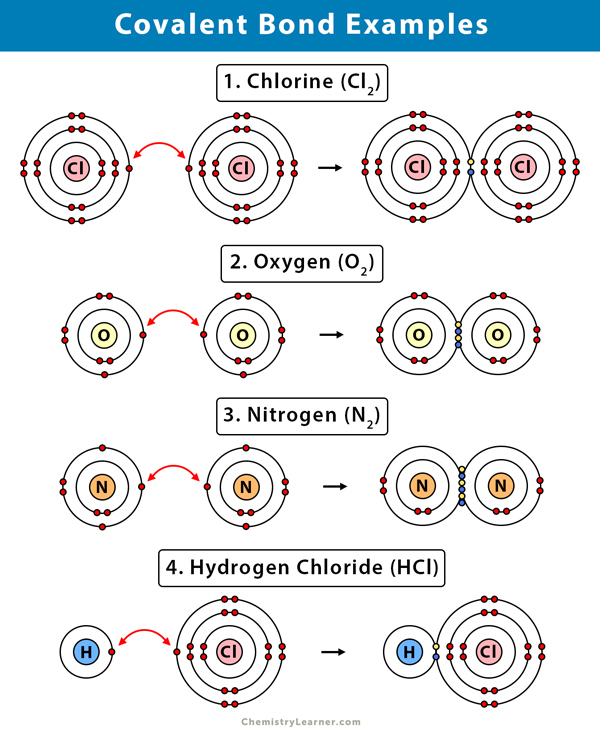
Which Compounds Would You Expect To Form A Covalent Bond - Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in. Structural isomers are compounds of the same chemical formula but different connectivities. Calcium is an alkaline earth metal in the second column of the periodic table. Is it possible to distinguish between ionic and. Lets take the ionic formula for calcium chloride, cacl_2. Compounds that contain carbon, hydrogen, and oxygen are organic. How do ionic compounds dissolve? How do ionic compounds conduct electricity in water?
Structural isomers are compounds of the same chemical formula but different connectivities. How do ionic compounds conduct electricity in water? In living things, organic compounds are often called biomolecules, but. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Calcium is an alkaline earth metal in the second column of the periodic table. Compounds that contain carbon, hydrogen, and oxygen are organic. Of course, not all ionic compounds are soluble in. Is it possible to distinguish between ionic and. How do ionic compounds dissolve? Lets take the ionic formula for calcium chloride, cacl_2.
Compounds that contain carbon, hydrogen, and oxygen are organic. How do ionic compounds dissolve? Is it possible to distinguish between ionic and. Lets take the ionic formula for calcium chloride, cacl_2. Structural isomers are compounds of the same chemical formula but different connectivities. How do ionic compounds conduct electricity in water? Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Calcium is an alkaline earth metal in the second column of the periodic table. In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in.
Covalent Bond Biology Dictionary
How do ionic compounds conduct electricity in water? Structural isomers are compounds of the same chemical formula but different connectivities. Calcium is an alkaline earth metal in the second column of the periodic table. Compounds that contain carbon, hydrogen, and oxygen are organic. Lets take the ionic formula for calcium chloride, cacl_2.
Covalent Bond Compound Examples, Formation Properties Video, 59 OFF
Lets take the ionic formula for calcium chloride, cacl_2. In living things, organic compounds are often called biomolecules, but. How do ionic compounds conduct electricity in water? Calcium is an alkaline earth metal in the second column of the periodic table. How do ionic compounds dissolve?
Covalent Bonding (Biology) — Definition & Role Expii
How do ionic compounds conduct electricity in water? How do ionic compounds dissolve? Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Is it possible to distinguish between ionic and. Of course, not all ionic compounds are soluble in.
Covalent Bond Definition and Examples
How do ionic compounds dissolve? Calcium is an alkaline earth metal in the second column of the periodic table. Structural isomers are compounds of the same chemical formula but different connectivities. How do ionic compounds conduct electricity in water? Because water is a solvent that is capable of solvating cations, and to a lesser extents anions.
Section 3 Covalent Bonds ppt download
Compounds that contain carbon, hydrogen, and oxygen are organic. How do ionic compounds conduct electricity in water? Structural isomers are compounds of the same chemical formula but different connectivities. Is it possible to distinguish between ionic and. How do ionic compounds dissolve?
Chapter 6 The Structure of Matter ppt download
Calcium is an alkaline earth metal in the second column of the periodic table. In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in. How do ionic compounds conduct electricity in water? Is it possible to distinguish between ionic and.
Covalent Bond Definition, Types, and Examples
Lets take the ionic formula for calcium chloride, cacl_2. How do ionic compounds conduct electricity in water? Calcium is an alkaline earth metal in the second column of the periodic table. How do ionic compounds dissolve? In living things, organic compounds are often called biomolecules, but.
Covalent Compound Examples
How do ionic compounds dissolve? Is it possible to distinguish between ionic and. Of course, not all ionic compounds are soluble in. Calcium is an alkaline earth metal in the second column of the periodic table. How do ionic compounds conduct electricity in water?
Covalent Compounds Examples and Properties
Calcium is an alkaline earth metal in the second column of the periodic table. How do ionic compounds dissolve? Compounds that contain carbon, hydrogen, and oxygen are organic. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Is it possible to distinguish between ionic and.
Reading Covalent Bonds Biology (Early Release)
Of course, not all ionic compounds are soluble in. Lets take the ionic formula for calcium chloride, cacl_2. Compounds that contain carbon, hydrogen, and oxygen are organic. Calcium is an alkaline earth metal in the second column of the periodic table. Is it possible to distinguish between ionic and.
In Living Things, Organic Compounds Are Often Called Biomolecules, But.
Is it possible to distinguish between ionic and. How do ionic compounds dissolve? Calcium is an alkaline earth metal in the second column of the periodic table. Lets take the ionic formula for calcium chloride, cacl_2.
How Do Ionic Compounds Conduct Electricity In Water?
Compounds that contain carbon, hydrogen, and oxygen are organic. Structural isomers are compounds of the same chemical formula but different connectivities. Of course, not all ionic compounds are soluble in. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions.