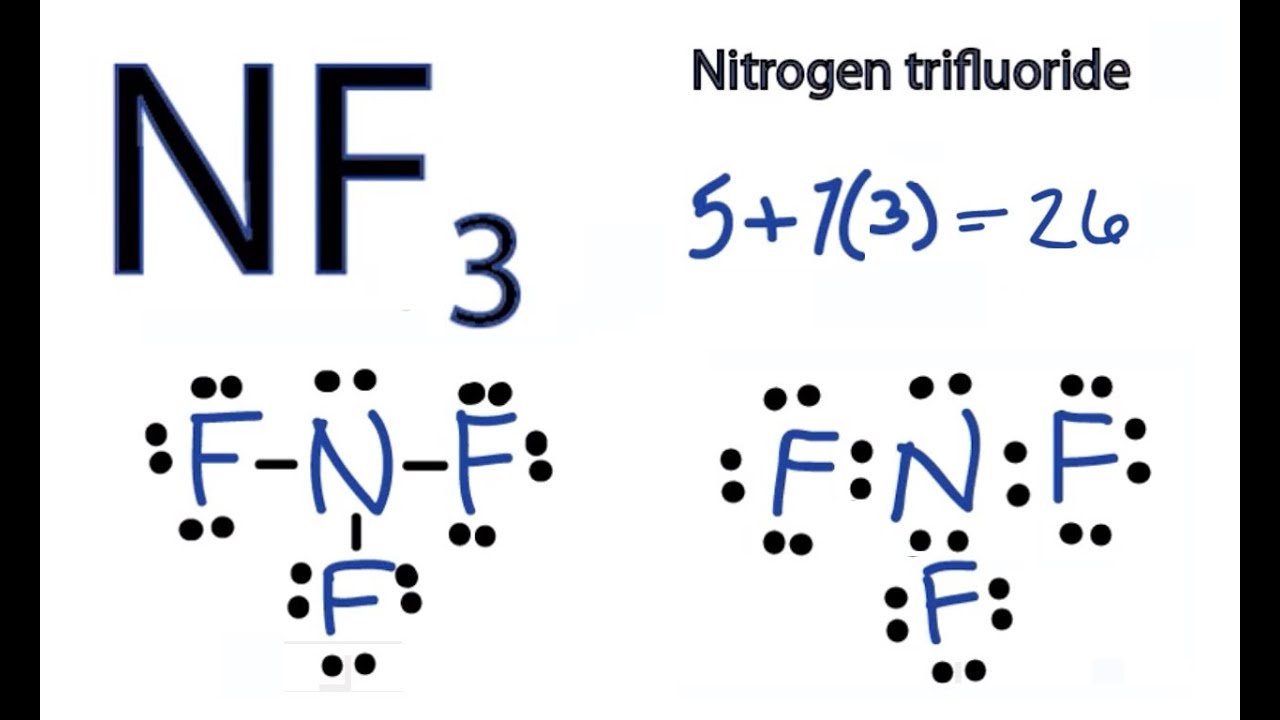
Nf3 Form - Wikipedia gives a better rundown,. Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. The knight develops to a good square that controls the center while keeping. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. 1.nf3 develops a piece instead of moving a pawn to begin the game. It's actually considered the main line. No, nf3 is not an ionic compound. Nf3 is a polar molecule.
It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. The knight develops to a good square that controls the center while keeping. 1.nf3 develops a piece instead of moving a pawn to begin the game. It's actually considered the main line. No, nf3 is not an ionic compound. Nf3 is a polar molecule. Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. Wikipedia gives a better rundown,. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven.
Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. Wikipedia gives a better rundown,. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. No, nf3 is not an ionic compound. Nf3 is a polar molecule. It's actually considered the main line. 1.nf3 develops a piece instead of moving a pawn to begin the game. The knight develops to a good square that controls the center while keeping.
NF3
Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. The knight develops to a good square that controls the center while keeping. Wikipedia gives a better rundown,. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. 1.nf3 develops.
Nf3 Form Fillable ≡ Fill Out Printable PDF Forms Online
No, nf3 is not an ionic compound. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. 1.nf3 develops a piece instead of moving a pawn to begin the game. Wikipedia gives a better rundown,. Nf3 is a polar molecule.
NYS Form NF3 ≡ Fill Out Printable PDF Forms Online
It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. Wikipedia gives a better rundown,. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven..
Unveiling the Lewis Dot Diagram for NF3 Understanding the Bonding
This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. The knight develops to a good square that controls the center while keeping. Nf3 is a polar molecule. No, nf3 is not an ionic compound. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds.
NF3
It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. The knight develops to a good square that controls the center while keeping. Nf3 is a polar molecule. Wikipedia gives a better rundown,.
Fillable Online nf3 form download Fax Email Print pdfFiller
Nf3 is a polar molecule. Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. 1.nf3 develops a piece instead of moving a pawn to begin the game. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. This is.
Nf 3 Form Fill Out and Sign Printable PDF Template airSlate SignNow
No, nf3 is not an ionic compound. It's actually considered the main line. Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. The knight develops to a good square that controls the center while keeping. 1.nf3 develops a piece instead of moving a pawn to begin.
Nitrogen Trifluorid Nf3 Molecule Royalty Free Vector Image, 59 OFF
The knight develops to a good square that controls the center while keeping. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds. Nf3 is a polar molecule. No, nf3 is not an ionic compound. This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven.
Draw The Lewis Structure Of The Molecule Nf3
Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. It's actually considered the main line. Wikipedia gives a better rundown,. The knight develops to a good square that controls the center while keeping. 1.nf3 develops a piece instead of moving a pawn to begin the game.
No Fault Nf3 20092025 Form Fill Out and Sign Printable PDF Template
Wikipedia gives a better rundown,. The knight develops to a good square that controls the center while keeping. It's actually considered the main line. 1.nf3 develops a piece instead of moving a pawn to begin the game. Nf3 is a polar molecule.
The Knight Develops To A Good Square That Controls The Center While Keeping.
Nf3, on the other hand, does not readily accept protons in water due to the presence of fluorine atoms that strongly attract electrons,. Nf3 is a polar molecule. 1.nf3 develops a piece instead of moving a pawn to begin the game. Wikipedia gives a better rundown,.
No, Nf3 Is Not An Ionic Compound.
This is because the electronegativity difference between nitrogen and fluorine atoms leads to an uneven. It's actually considered the main line. It is a covalent compound where nitrogen (n) and fluorine (f) atoms share electrons to form bonds.