Ivdr Performance Evaluation Report Template - The contents of two new ivdr documents are explicitly described in annex xiii: Get an ivdr performance evaluation report template for medical devices. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. The performance evaluation plan (pep) and the performance. The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. Learn how to assess device compliance with ivdr guidelines effectively
The performance evaluation plan (pep) and the performance. Get an ivdr performance evaluation report template for medical devices. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. The contents of two new ivdr documents are explicitly described in annex xiii: If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. Learn how to assess device compliance with ivdr guidelines effectively The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate.
This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. Get an ivdr performance evaluation report template for medical devices. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. The performance evaluation plan (pep) and the performance. The contents of two new ivdr documents are explicitly described in annex xiii: Learn how to assess device compliance with ivdr guidelines effectively
Performance Evaluation Report Sample → Free Report Examples
This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. Learn how to assess device compliance with ivdr guidelines effectively The performance evaluation plan (pep) and the performance..
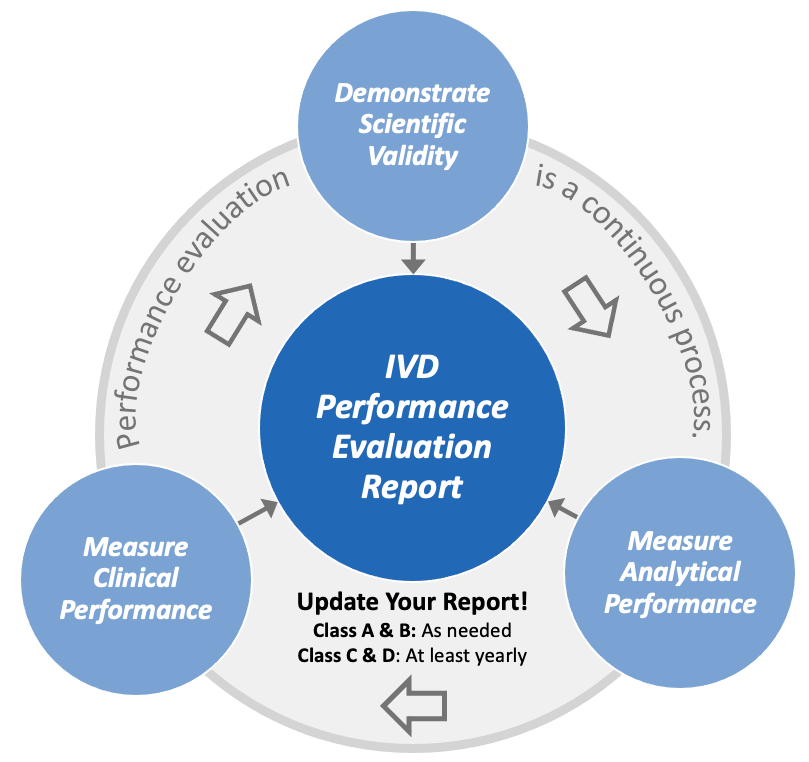
Performance plan evaluation under the EU IVDR 2017/746
Learn how to assess device compliance with ivdr guidelines effectively The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. The performance evaluation plan (pep) and the performance..
IVDR Practical Considerations for the Performance Evaluation Plan and
The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. Learn how to assess device compliance with ivdr guidelines effectively Get an ivdr performance evaluation report template for medical devices. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance.
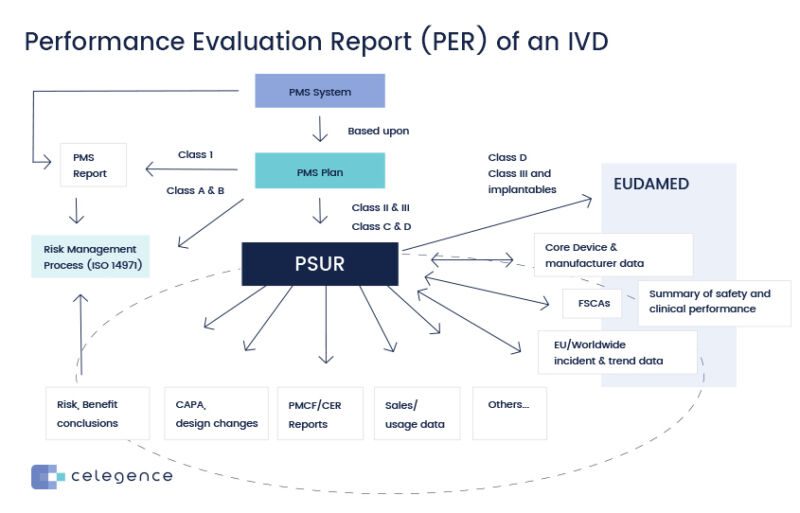
IVDR Performance Evaluation Report Oriel STAT A MATRIX
This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. Learn how to assess device compliance with ivdr guidelines effectively The contents of two new.
PPT EU IVDR Performance Validation Report Steps and Requirements
If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. The performance evaluation plan (pep) and the performance. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. Learn how to assess device compliance with.
Performance Evaluation Under IVDR Proving IVD Performance
Learn how to assess device compliance with ivdr guidelines effectively Get an ivdr performance evaluation report template for medical devices. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. The performance evaluation plan (pep) and the performance. The contents of two new ivdr documents.
Ensuring Compliance for your IVD's Performance Evaluation Part 1
Learn how to assess device compliance with ivdr guidelines effectively The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. Get an ivdr performance evaluation report template for medical devices. If you are a user of formwork, our eqms software, you can save a lot of time by.
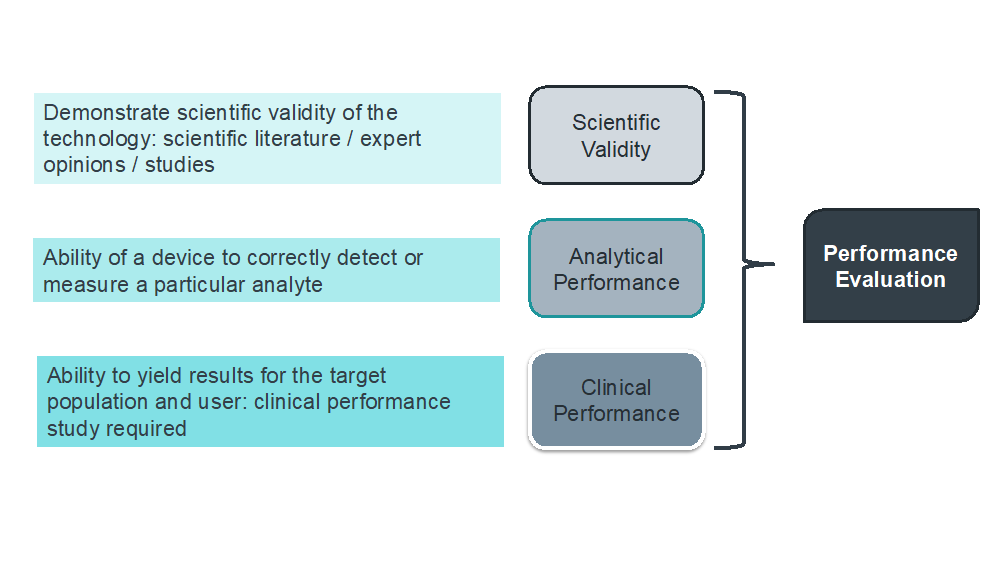
Clinical evidence requirements according to the IVDR 2017/746
Get an ivdr performance evaluation report template for medical devices. The performance evaluation plan shall specify the characteristics and the performance of the device and the process and criteria applied to generate. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. This document outlines.
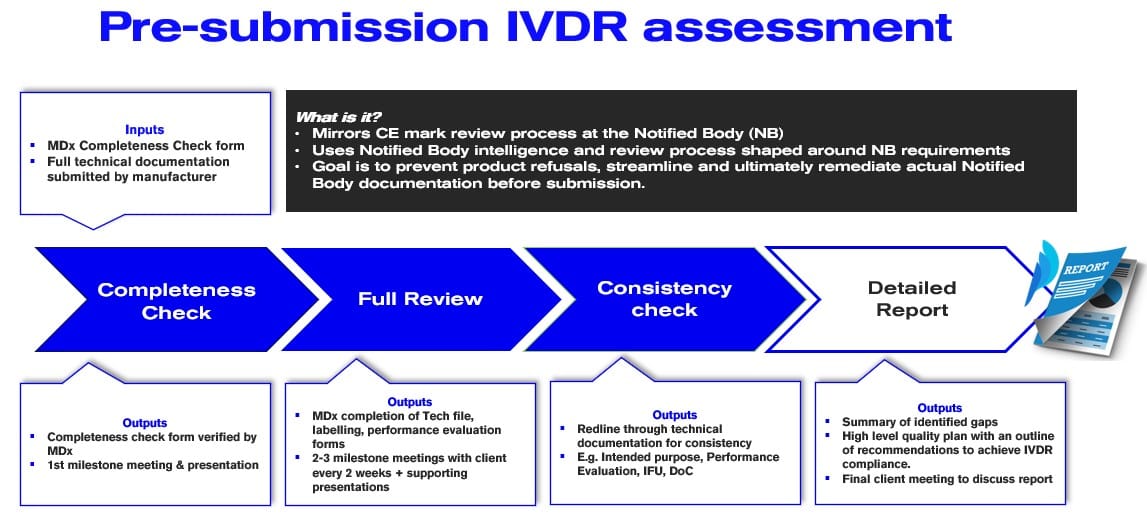
IVDR Compliance Made Easy The Importance of an IVDR PreSubmission
If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. Get an ivdr performance evaluation report template for medical devices. Learn how to assess device.
IVDR Performance Evaluation Report Template
Learn how to assess device compliance with ivdr guidelines effectively This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. The contents of two new ivdr documents are explicitly described in annex xiii: Get an ivdr performance evaluation report template for medical devices. The performance evaluation plan shall specify the.
The Performance Evaluation Plan Shall Specify The Characteristics And The Performance Of The Device And The Process And Criteria Applied To Generate.
This document outlines the general principles of clinical evidence and provides guidance on the continuous process of performance evaluation for. Get an ivdr performance evaluation report template for medical devices. If you are a user of formwork, our eqms software, you can save a lot of time by choosing “qms” on the top menu and. Learn how to assess device compliance with ivdr guidelines effectively
The Performance Evaluation Plan (Pep) And The Performance.
The contents of two new ivdr documents are explicitly described in annex xiii: