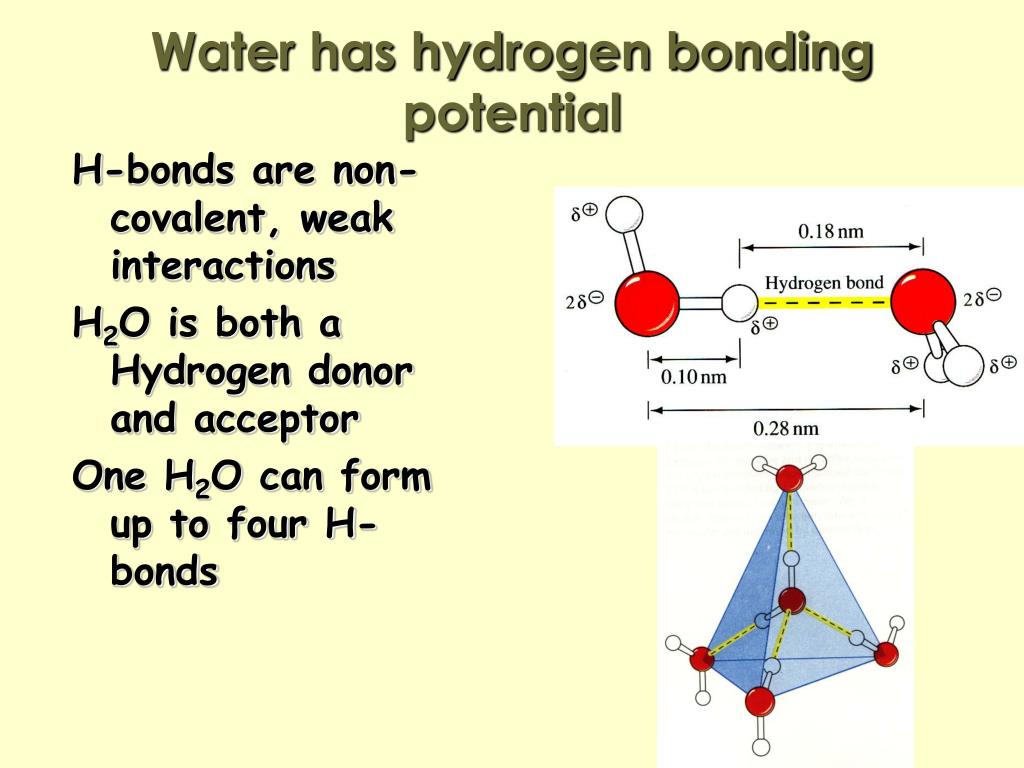
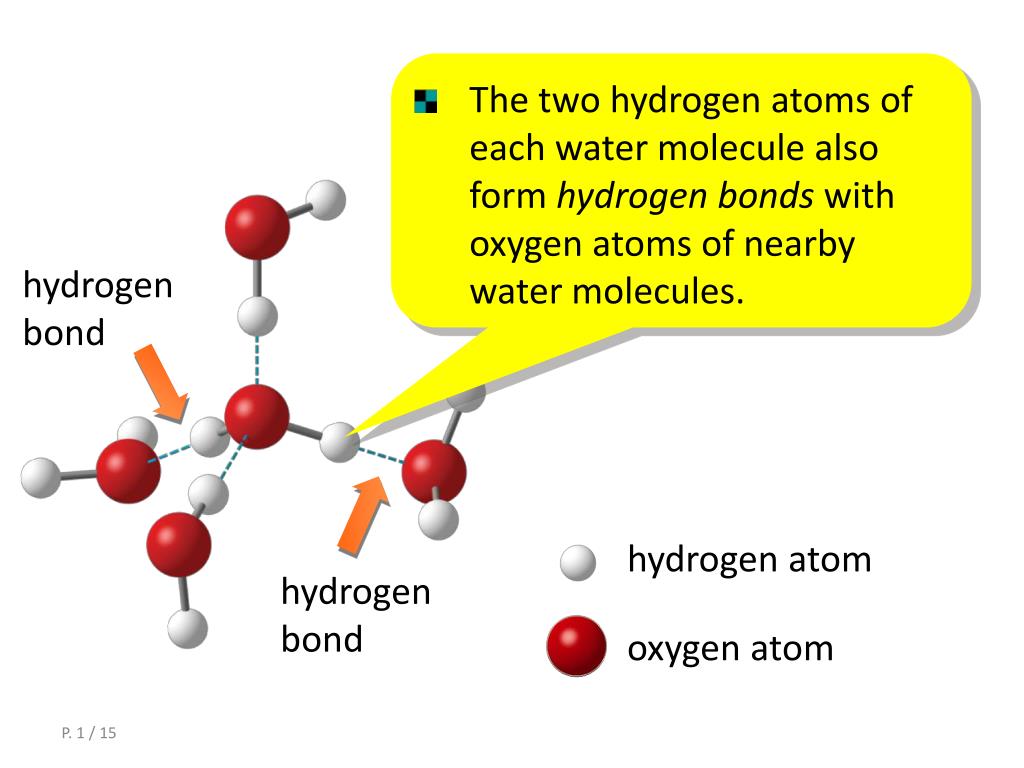
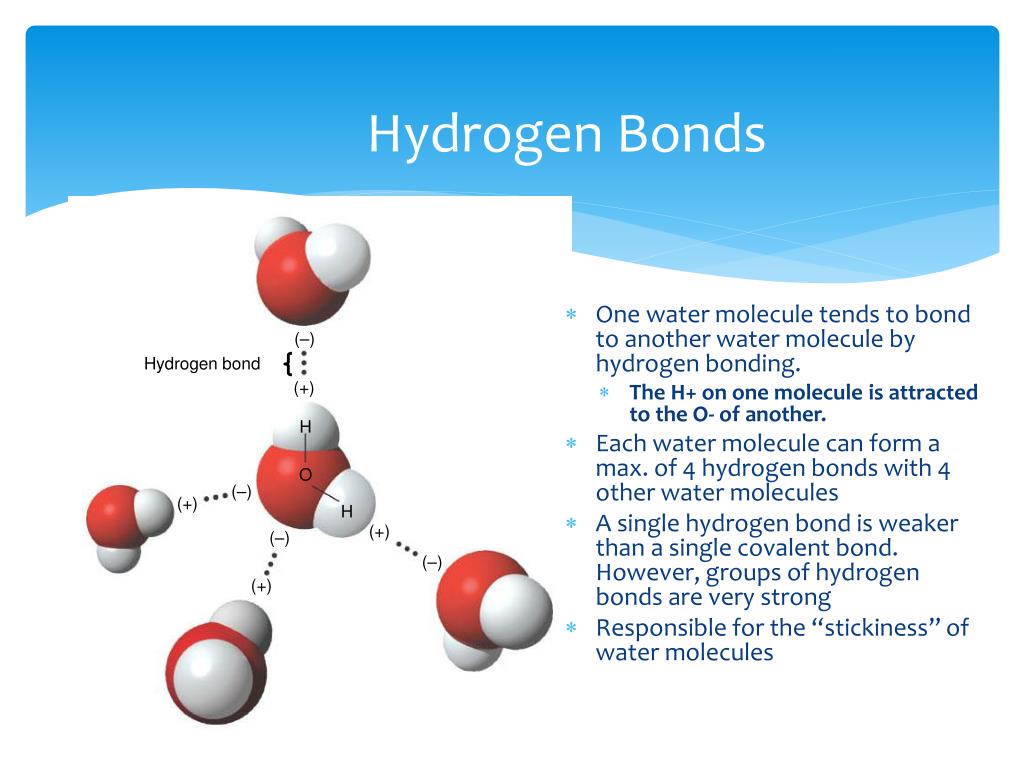
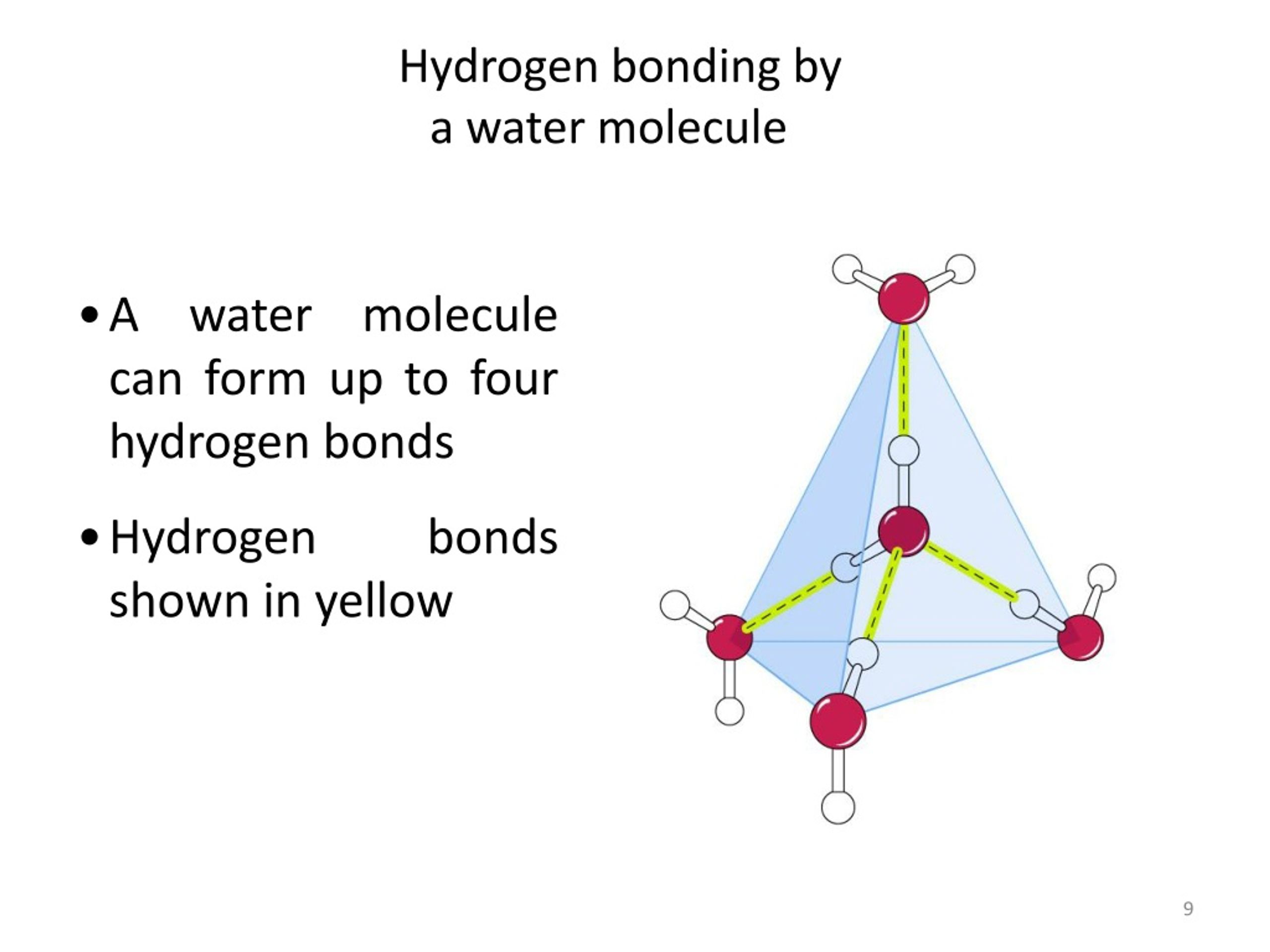
How Many Hydrogen Bonds Can Water Form - Water is made up of one oxygen atom covalently bonded to. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. The negative end of one. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. A hydrogen bond is the weak electrostatic attraction between different molecules such as water.
Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. A hydrogen bond is the weak electrostatic attraction between different molecules such as water. Water is made up of one oxygen atom covalently bonded to. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. The negative end of one. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the.
A hydrogen bond is the weak electrostatic attraction between different molecules such as water. Water is made up of one oxygen atom covalently bonded to. The negative end of one. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds.
Unit 2 (Biochemistry) Notes, Part 1 Atomic And Molecular Structure
Water is made up of one oxygen atom covalently bonded to. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. The negative end of.
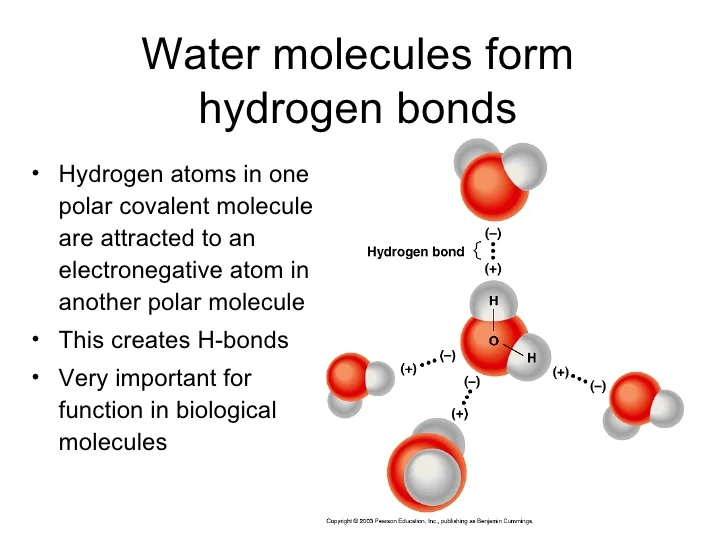
Biology Chapter ppt download
A hydrogen bond is the weak electrostatic attraction between different molecules such as water. The negative end of one. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one.
Overview A Chemical Connection to Biology ppt download
7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. A hydrogen bond is the weak electrostatic attraction between different molecules such as water. The negative end of one. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another..
PPT Properties of water PowerPoint Presentation, free download ID
A hydrogen bond is the weak electrostatic attraction between different molecules such as water. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. The negative end of one. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it.
How Water Forms Hydrogen Bonds YouTube
Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. The negative end of one. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Water is made up of one oxygen atom covalently bonded to. Water is.
PPT hydrogen bond PowerPoint Presentation, free download ID4524678
Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Water is made up of one oxygen atom covalently bonded to. Water is capable of participating in 4.
Hydrogen Bonds Between Water Molecules Diagram Science Onlin
Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Water is capable of participating in 4 hydrogen bonds at once, granted it only does this when it forms a perfect crystal structure. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it.
PPT Lec.1 Chemistry Of Water PowerPoint Presentation, free download
Water is made up of one oxygen atom covalently bonded to. The negative end of one. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. A hydrogen.
Water Review
Water is made up of one oxygen atom covalently bonded to. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. The negative end of one. Hydrogen bonds hydrogen bonds form when two polar molecules (one of them being hydrogen) are attracted to one another. Theoretically, there are.
Hydrogen Bonds — Overview & Examples Expii
7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the. Water is made up of one oxygen atom covalently bonded to. Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. A hydrogen bond is.
Water Is Capable Of Participating In 4 Hydrogen Bonds At Once, Granted It Only Does This When It Forms A Perfect Crystal Structure.
Theoretically, there are a maximum of 5 water molecules that one urea molecule can hydrogen bond with, but there are 6 hydrogen bonds. The negative end of one. A hydrogen bond is the weak electrostatic attraction between different molecules such as water. 7 we usually get some questions about how many hydrogen bonds these compounds can create, we use it to differentiate between the.
Hydrogen Bonds Hydrogen Bonds Form When Two Polar Molecules (One Of Them Being Hydrogen) Are Attracted To One Another.
Water is made up of one oxygen atom covalently bonded to.