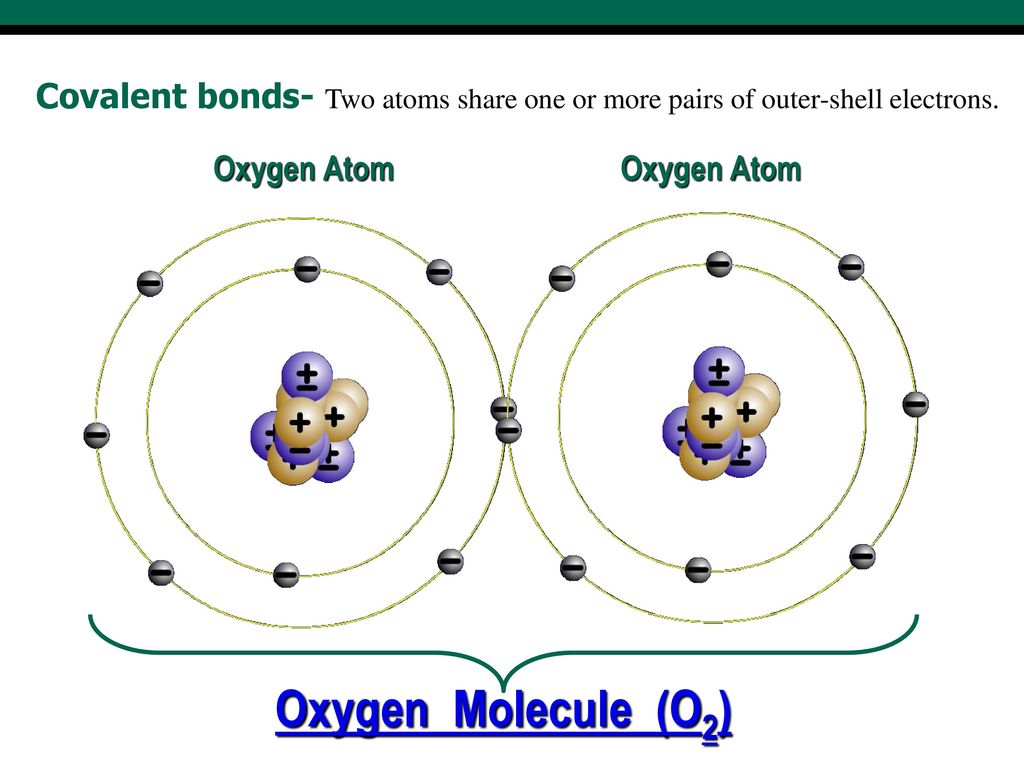
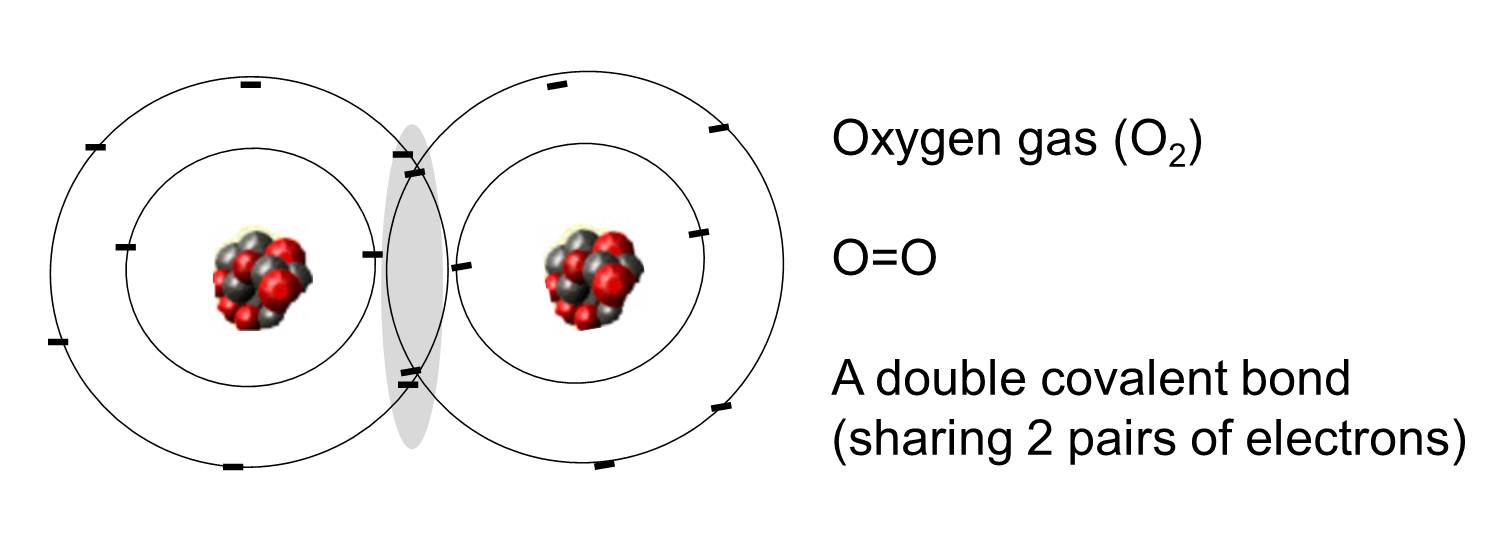
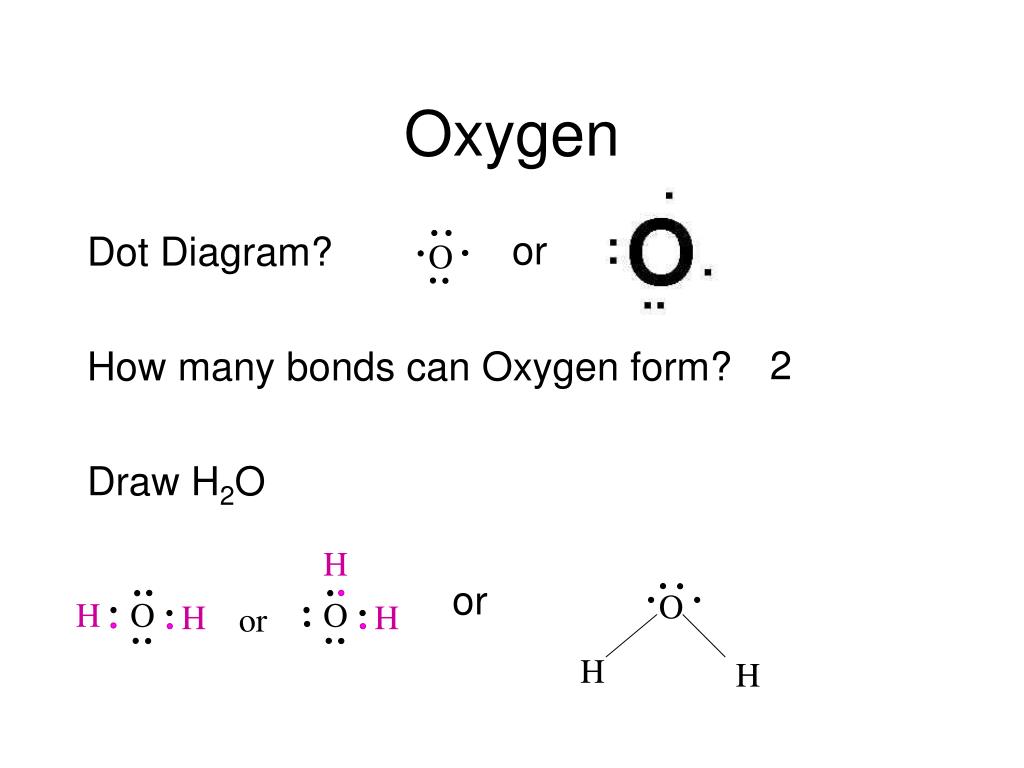
How Many Covalent Bonds Can Oxygen Form - But why does this happen? This is because oxygen normally forms two covalent. The chemical bond for oxygen is primarily the covalent bond. And that will most likely be. How many covalent bonds can be formed from an atom? 13 coordinate covalent bonds are bonds on which both electrons from one atom. Some may think it is because one of the. Oxygen atoms tend to form double bonds with each other, sharing. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. An oxygen atom with a negative charge can form two covalent bonds.
Oxygen atoms tend to form double bonds with each other, sharing. But why does this happen? It depends on the element following what one might call normal valence. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. 13 coordinate covalent bonds are bonds on which both electrons from one atom. Some may think it is because one of the. This is because oxygen normally forms two covalent. An oxygen atom with a negative charge can form two covalent bonds. The chemical bond for oxygen is primarily the covalent bond. How many covalent bonds can be formed from an atom?
An oxygen atom with a negative charge can form two covalent bonds. How many covalent bonds can be formed from an atom? In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. Some may think it is because one of the. It depends on the element following what one might call normal valence. The chemical bond for oxygen is primarily the covalent bond. This is because oxygen normally forms two covalent. But why does this happen? Oxygen atoms tend to form double bonds with each other, sharing. 13 coordinate covalent bonds are bonds on which both electrons from one atom.
PPT Structure and Bonding PowerPoint Presentation, free download ID
The chemical bond for oxygen is primarily the covalent bond. This is because oxygen normally forms two covalent. But why does this happen? Some may think it is because one of the. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration.
Covalent bonding in an oxygen molecule. Chemistry Activities, Gcse
Some may think it is because one of the. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. But why does this happen? The chemical bond for oxygen is primarily the covalent bond. 13 coordinate covalent bonds are bonds on which both electrons from one atom.
Biology I Covalent Bonds ppt download
Oxygen atoms tend to form double bonds with each other, sharing. How many covalent bonds can be formed from an atom? It depends on the element following what one might call normal valence. The chemical bond for oxygen is primarily the covalent bond. Some may think it is because one of the.
Covalent Bonding In An Oxygen Molecule vrogue.co
It depends on the element following what one might call normal valence. And that will most likely be. An oxygen atom with a negative charge can form two covalent bonds. Oxygen atoms tend to form double bonds with each other, sharing. This is because oxygen normally forms two covalent.
Covalent Bond A Level Chemistry Revision Notes
But why does this happen? It depends on the element following what one might call normal valence. Some may think it is because one of the. 13 coordinate covalent bonds are bonds on which both electrons from one atom. How many covalent bonds can be formed from an atom?
PPT Chemistry Bonding PowerPoint Presentation, free download ID
Some may think it is because one of the. The chemical bond for oxygen is primarily the covalent bond. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. This is because oxygen normally forms two covalent. And that will most likely be.
Primary and Secondary Bonds Owlcation
But why does this happen? This is because oxygen normally forms two covalent. An oxygen atom with a negative charge can form two covalent bonds. The chemical bond for oxygen is primarily the covalent bond. Some may think it is because one of the.
PPT KS4 Chemistry PowerPoint Presentation, free download ID2755080
And that will most likely be. It depends on the element following what one might call normal valence. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. But why does this happen? An oxygen atom with a negative charge can form two covalent bonds.
Notes Covalent Bonds 9/24/18 ppt download
Some may think it is because one of the. Oxygen atoms tend to form double bonds with each other, sharing. It depends on the element following what one might call normal valence. In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. 13 coordinate covalent bonds are bonds on.
Covalent Bonding GCSE Chemistry Revision
But why does this happen? In general your assumption is correct, that it is possible to form only three covalent bonds to reach a stable configuration. And that will most likely be. Oxygen atoms tend to form double bonds with each other, sharing. 13 coordinate covalent bonds are bonds on which both electrons from one atom.
An Oxygen Atom With A Negative Charge Can Form Two Covalent Bonds.
Some may think it is because one of the. And that will most likely be. It depends on the element following what one might call normal valence. How many covalent bonds can be formed from an atom?
13 Coordinate Covalent Bonds Are Bonds On Which Both Electrons From One Atom.
The chemical bond for oxygen is primarily the covalent bond. This is because oxygen normally forms two covalent. Oxygen atoms tend to form double bonds with each other, sharing. But why does this happen?