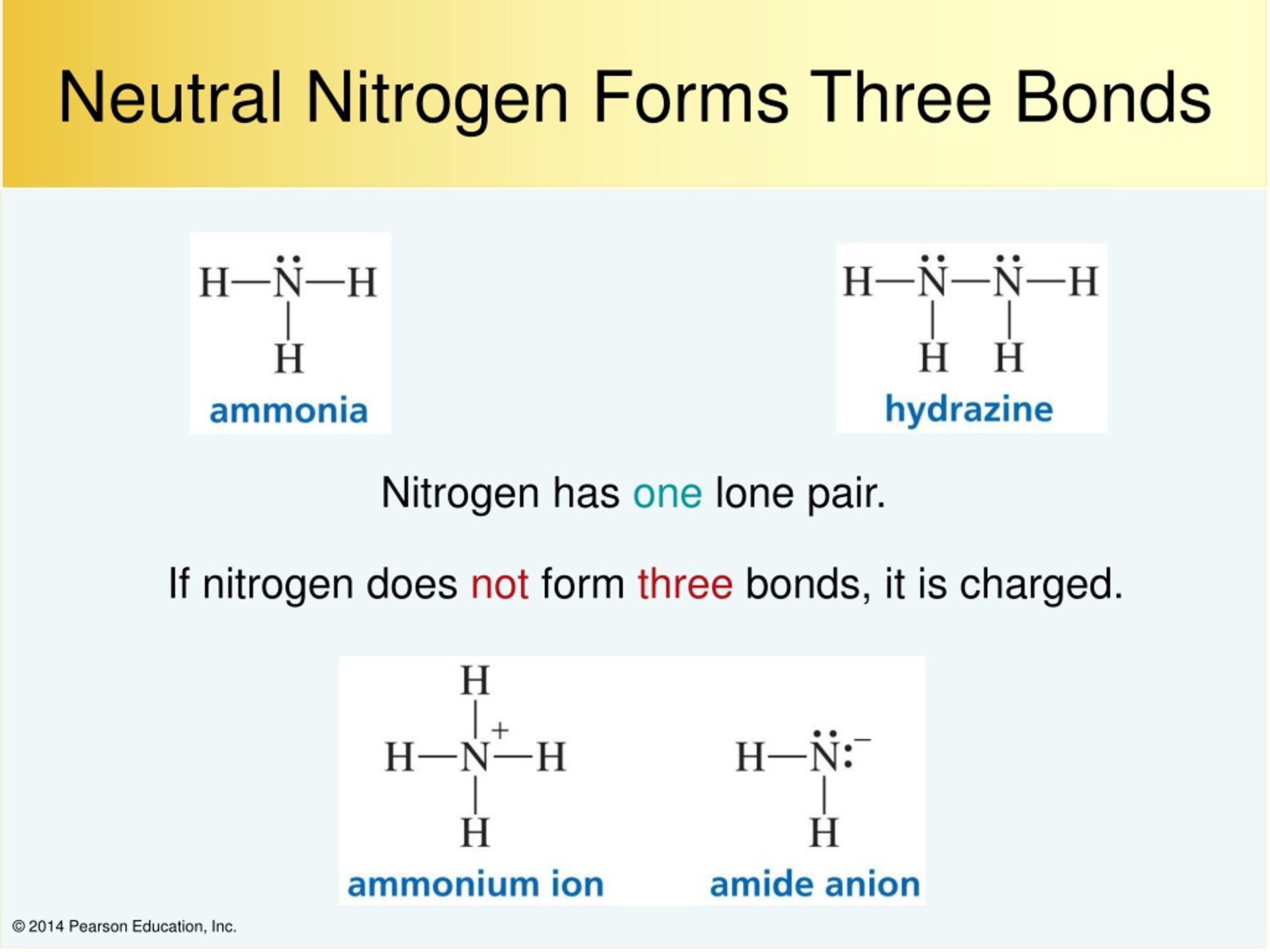
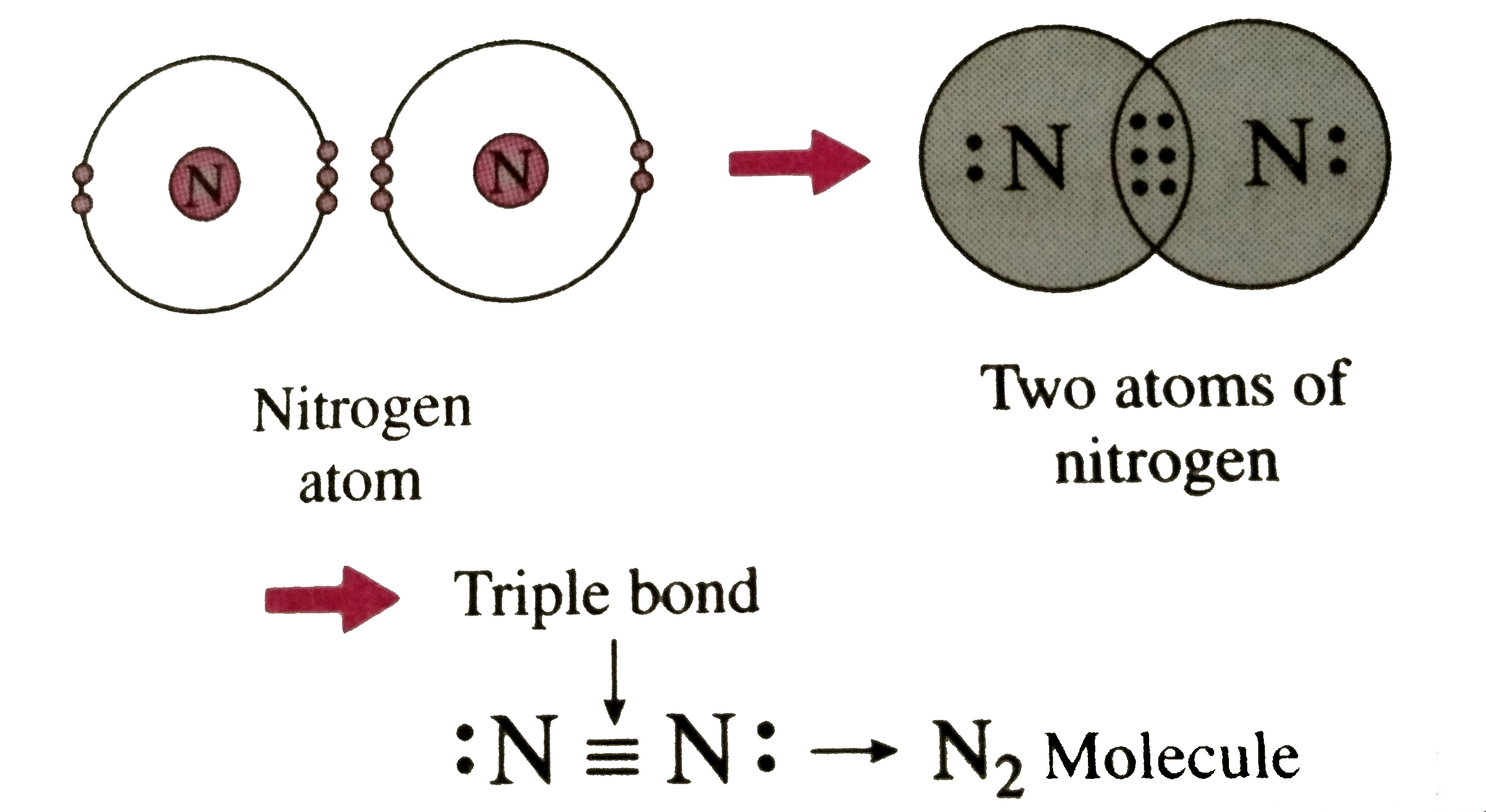How Many Covalent Bonds Can Nitrogen Form - Ammonia is a nitrogen atom bonded to three hydrogen atoms. In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. It has five valence electrons and tends to share three electrons with other atoms to. Nitrogen normally forms three covalent bonds. There is a total of three covalent. It is one of the main components of air. How many covalent bonds are in ammonia? These electrons are located in the valence shell. The exact location of nitrogen in the periodic table is the second period and 15th group.
It is one of the main components of air. Ammonia is a nitrogen atom bonded to three hydrogen atoms. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. How many covalent bonds are in ammonia? It has five valence electrons and tends to share three electrons with other atoms to. There is a total of three covalent. Nitrogen normally forms three covalent bonds. In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. The exact location of nitrogen in the periodic table is the second period and 15th group. These electrons are located in the valence shell.
In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. It is one of the main components of air. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. The exact location of nitrogen in the periodic table is the second period and 15th group. How many covalent bonds are in ammonia? Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent. It has five valence electrons and tends to share three electrons with other atoms to. Nitrogen normally forms three covalent bonds. These electrons are located in the valence shell.
Covalent bonding
It is one of the main components of air. Nitrogen normally forms three covalent bonds. In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. It has five valence electrons and tends to share three electrons with other atoms to. The exact location of nitrogen in the periodic table.
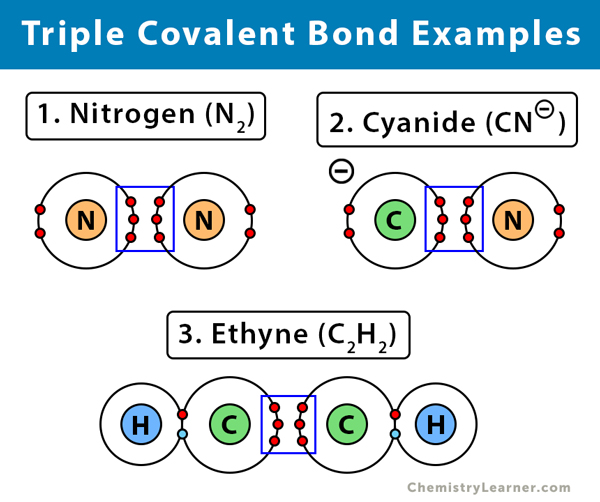
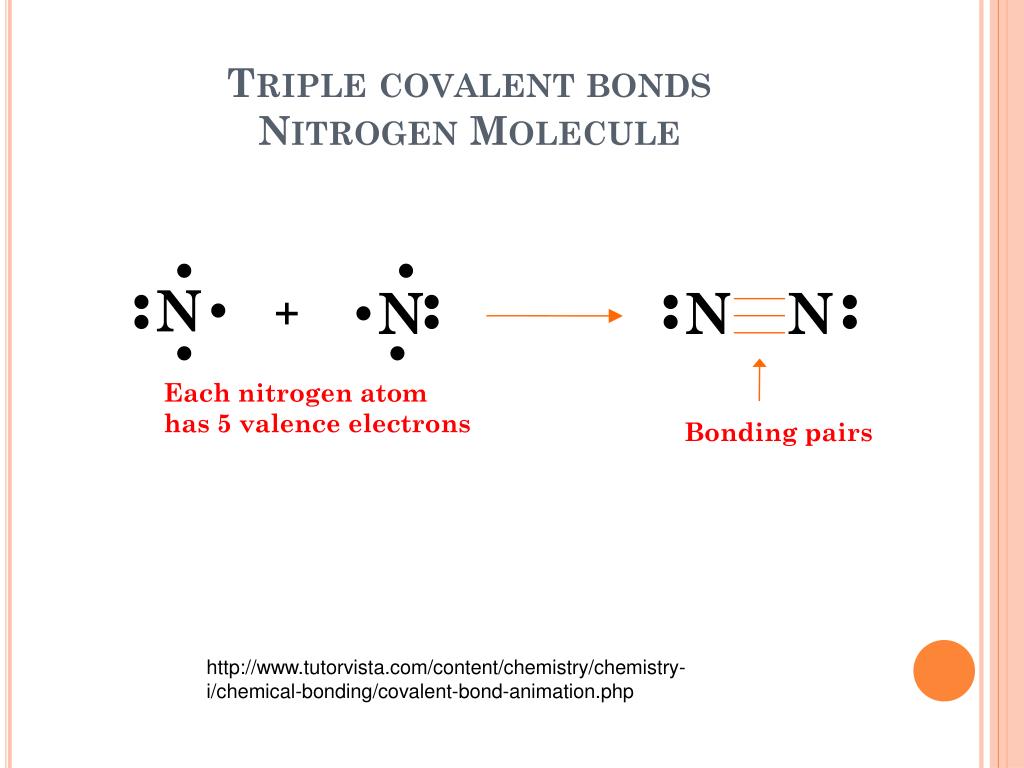
Nitrogen Molecule Covalent Bond
A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. It is one of the main components of air. Ammonia is a nitrogen atom bonded to three hydrogen atoms. These electrons are located in the valence shell. In order to achieve a stable octet configuration, nitrogen can share three electrons with other.
PPT Covalent Bonding and Chemical Bonds PowerPoint Presentation, free
These electrons are located in the valence shell. In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. Ammonia is a nitrogen atom bonded to three hydrogen atoms. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. It is one.
PPT COVALENT BONDING PowerPoint Presentation, free download ID5128236
It has five valence electrons and tends to share three electrons with other atoms to. The exact location of nitrogen in the periodic table is the second period and 15th group. It is one of the main components of air. Nitrogen normally forms three covalent bonds. These electrons are located in the valence shell.
Covalent Bond N2
It has five valence electrons and tends to share three electrons with other atoms to. It is one of the main components of air. The exact location of nitrogen in the periodic table is the second period and 15th group. How many covalent bonds are in ammonia? In order to achieve a stable octet configuration, nitrogen can share three electrons.
PPT Remembering General Chemistry Electronic Structure and Bonding
In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. These electrons are located in the valence shell. It is one of the main components of air. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. There is a total.
PPT KS4 Chemistry PowerPoint Presentation, free download ID2755080
These electrons are located in the valence shell. It has five valence electrons and tends to share three electrons with other atoms to. There is a total of three covalent. Ammonia is a nitrogen atom bonded to three hydrogen atoms. How many covalent bonds are in ammonia?
Nitrogen Covalent Bond
A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. Ammonia is a nitrogen atom bonded to three hydrogen atoms. In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. These electrons are located in the valence shell. How many covalent.
Covalent Bonding Chemistry Lesson 1. ppt video online download
Nitrogen normally forms three covalent bonds. Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent. It has five valence electrons and tends to share three electrons with other atoms to. These electrons are located in the valence shell.
Describe the formation of nitrogen molecule.
In order to achieve a stable octet configuration, nitrogen can share three electrons with other atoms to form three covalent bonds,. There is a total of three covalent. A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. The exact location of nitrogen in the periodic table is the second period and.
Ammonia Is A Nitrogen Atom Bonded To Three Hydrogen Atoms.
A covalent bond is a bond between two atoms in which electrons are shared to form electron pairs. These electrons are located in the valence shell. There is a total of three covalent. It has five valence electrons and tends to share three electrons with other atoms to.
In Order To Achieve A Stable Octet Configuration, Nitrogen Can Share Three Electrons With Other Atoms To Form Three Covalent Bonds,.
It is one of the main components of air. The exact location of nitrogen in the periodic table is the second period and 15th group. Nitrogen normally forms three covalent bonds. How many covalent bonds are in ammonia?