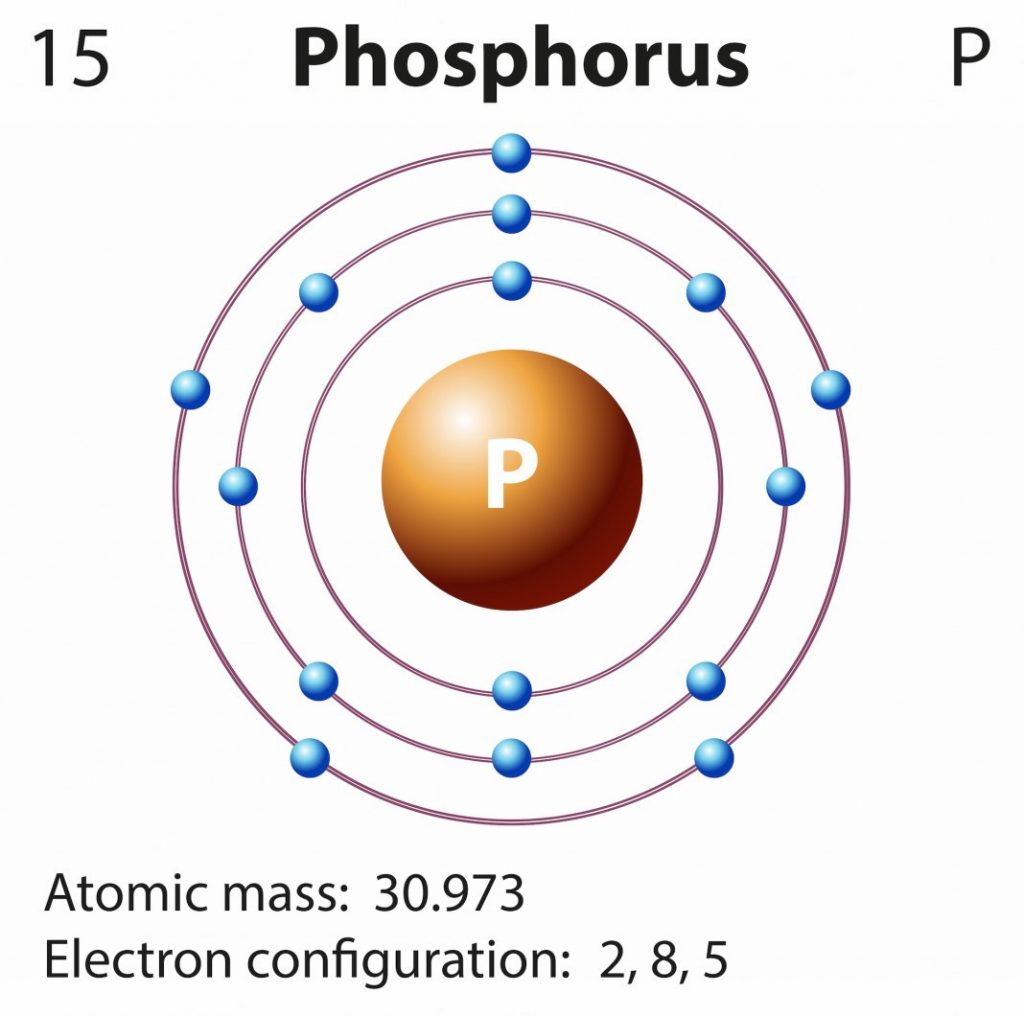
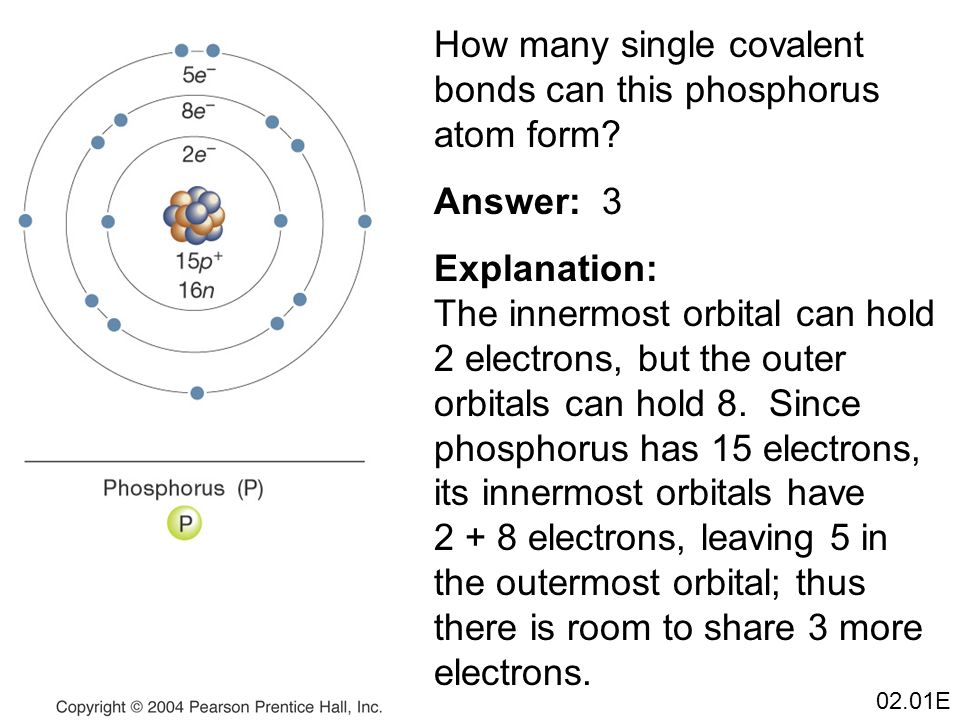
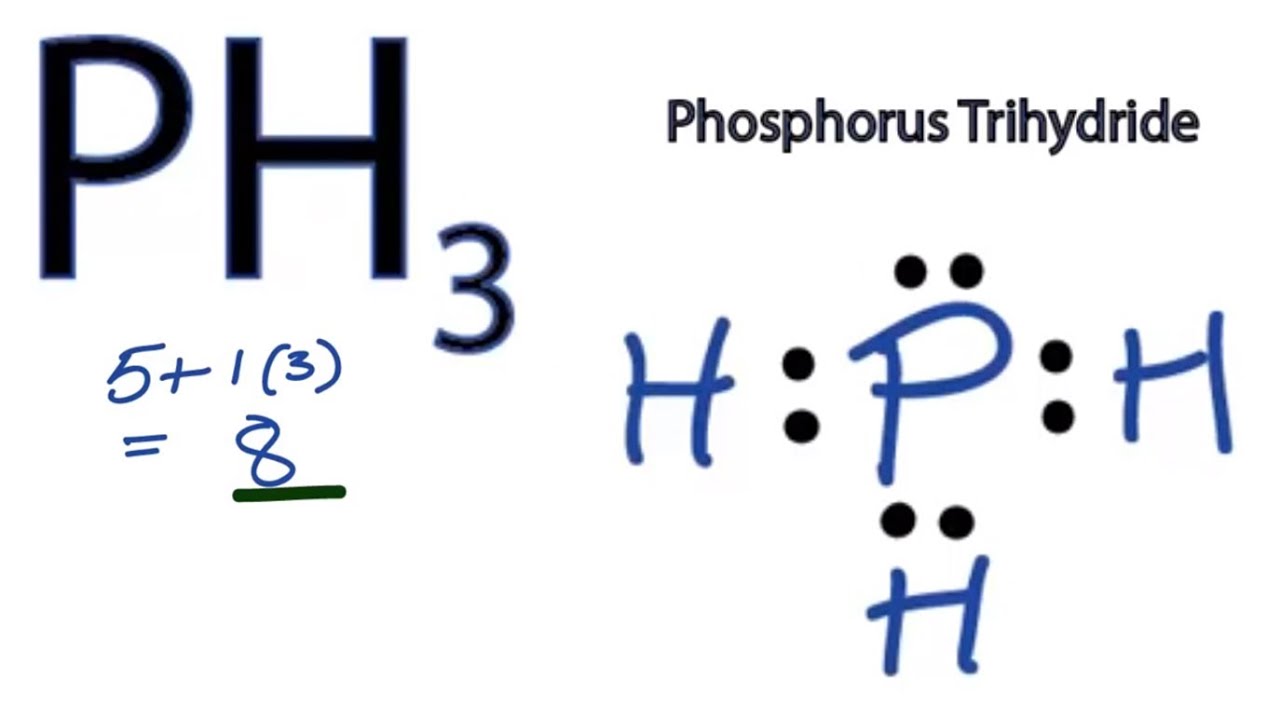
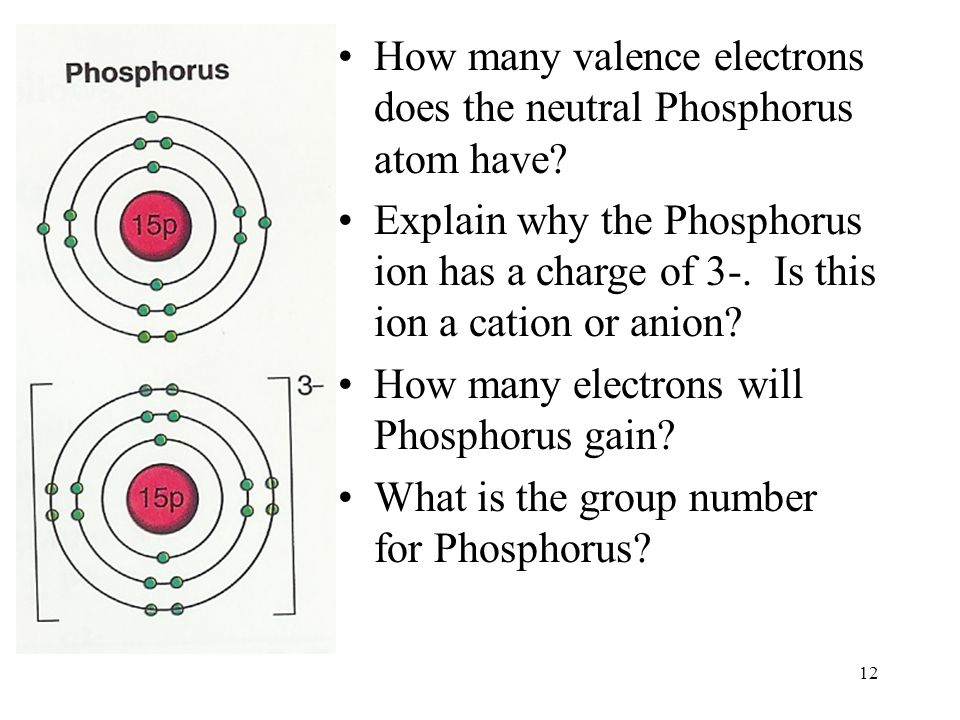
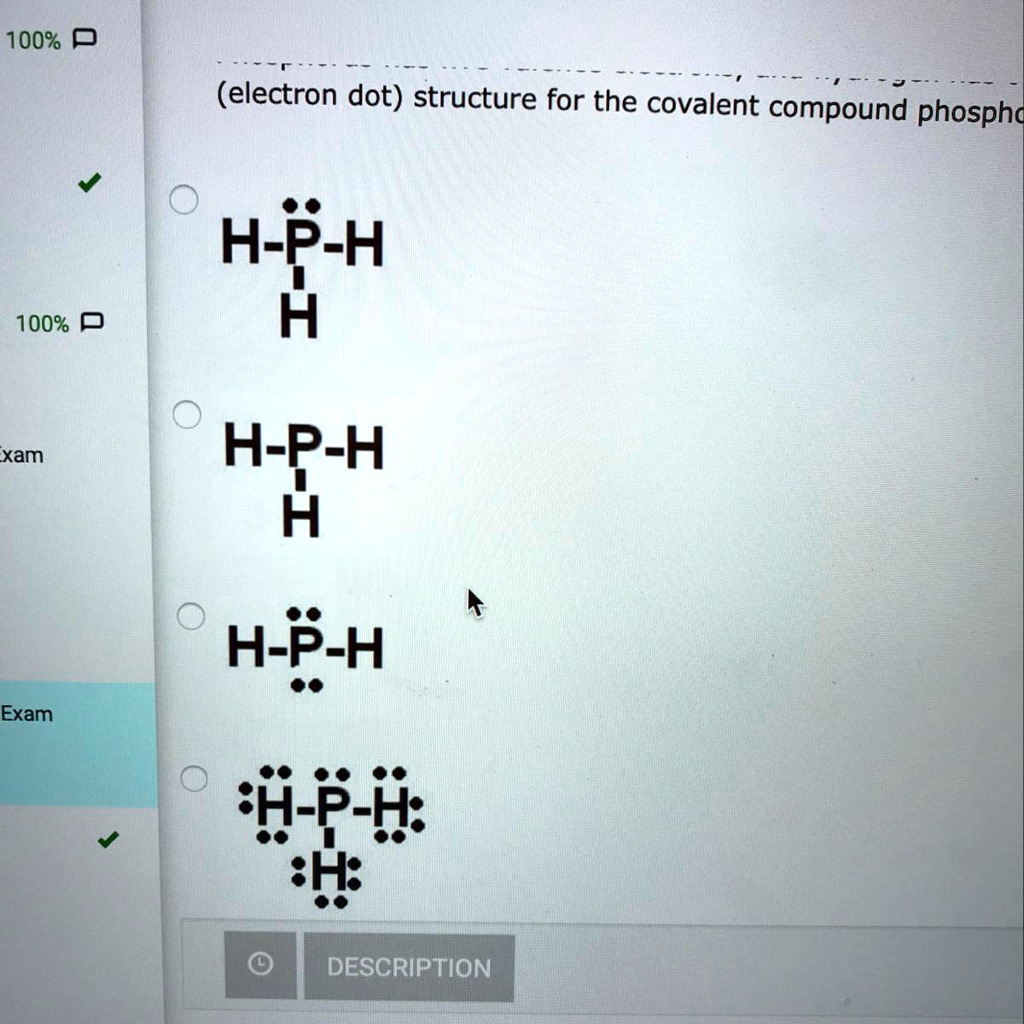
How Many Bonds Does Phosphorus Form - It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. For a fourth bond to form, another atom would have to donate two electrons to the bond. This leads to the formation of. This happens in many cases, such as when an amine reacts. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. Phosphorus typically forms 3 covalent bonds. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form.
I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. This happens in many cases, such as when an amine reacts. Phosphorus typically forms 3 covalent bonds. This leads to the formation of. For a fourth bond to form, another atom would have to donate two electrons to the bond.
For a fourth bond to form, another atom would have to donate two electrons to the bond. This leads to the formation of. This happens in many cases, such as when an amine reacts. Phosphorus typically forms 3 covalent bonds. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion:
Phosphorus Definition, Facts, Symbol, Discovery, Property, Uses
For a fourth bond to form, another atom would have to donate two electrons to the bond. Phosphorus typically forms 3 covalent bonds. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. This leads to the formation of. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and.
Phosphorus Atom Model Project
This happens in many cases, such as when an amine reacts. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: This leads to the formation of. Phosphorus typically forms 3 covalent bonds. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons.
Phosphate Ion Functional Groups Biochemistry Vector Infographic Stock
I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus typically forms 3 covalent bonds. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. This leads to the.
Chapter 8 Covalent Bonding Covalent bonding Usually forms
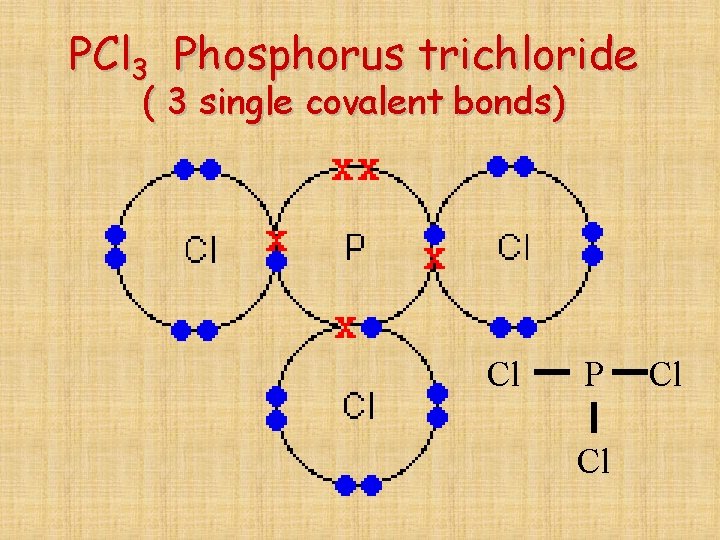
Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. This leads to the formation of. Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. For a fourth bond to form, another atom would have to donate two electrons to the bond. It.
Po43 P
Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. For a fourth bond to form, another atom would have to donate two electrons to the bond. This happens in many cases, such as.
Lewis Dot Diagram Phosphorus Wiring Diagram Pictures
This leads to the formation of. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. Phosphorus typically forms 3 covalent bonds.
Phosphorus Orbital diagram, Electron configuration, and Valence electrons
It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. This happens in many cases, such as when an amine reacts. This leads to the formation of. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. For a fourth bond to form, another atom.
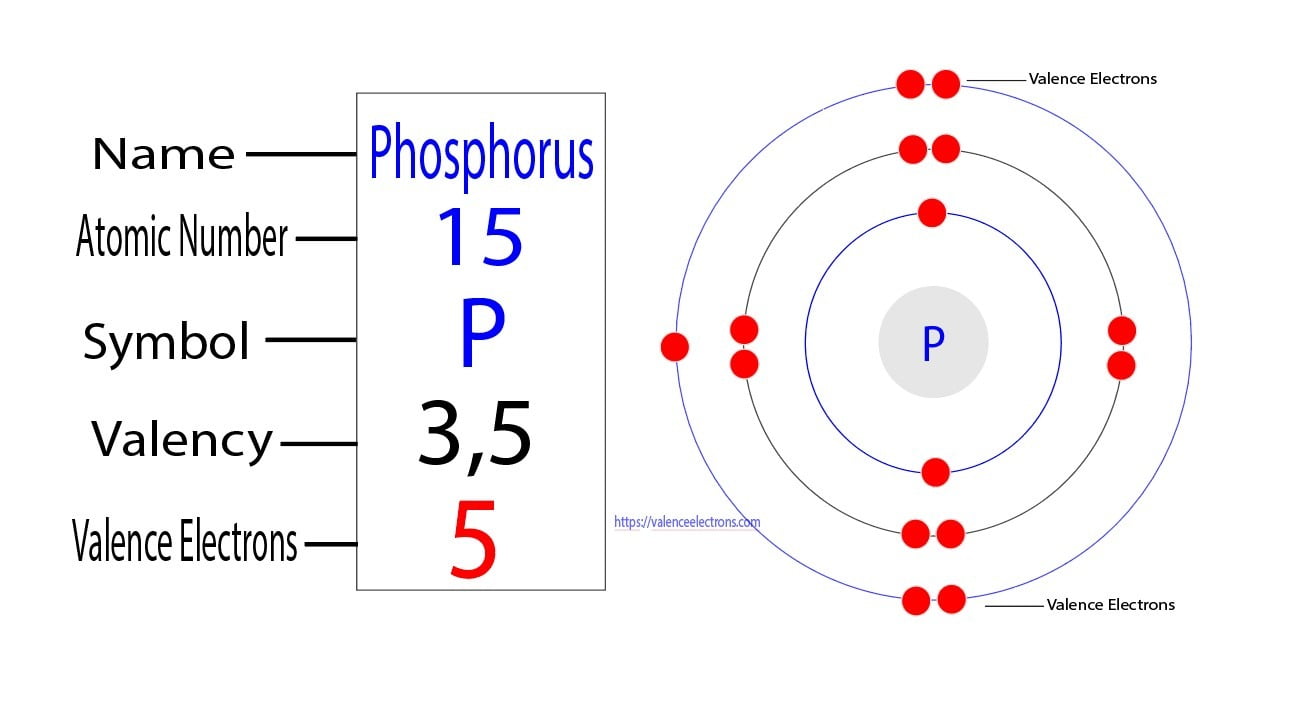
Phosphorus Valence Electrons Phosphorus Valency (P) with Dot Diagram
Phosphorus typically forms 3 covalent bonds. This happens in many cases, such as when an amine reacts. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: For a fourth bond to form, another atom would have to donate two electrons.
Phosphorus Lewis Dot Diagram
This leads to the formation of. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus typically forms 3 covalent bonds. I'm looking for an explanation of the bonding in the phosphate (po 43−).
What Are The Different Industrial Uses Of Phosphorus
This happens in many cases, such as when an amine reacts. I'm looking for an explanation of the bonding in the phosphate (po 43−) ion: Phosphorus typically forms 3 covalent bonds. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form. Phosphorus (p) typically forms 3 covalent bonds due to.
I'm Looking For An Explanation Of The Bonding In The Phosphate (Po 43−) Ion:
Phosphorus (p) typically forms 3 covalent bonds due to its electronic configuration of 5 valence electrons. This happens in many cases, such as when an amine reacts. It has 5 valence electrons and can achieve a stable electron configuration by sharing electrons. For a fourth bond to form, another atom would have to donate two electrons to the bond.
This Leads To The Formation Of.
Phosphorus typically forms 3 covalent bonds. Yes, phosphorus trichloride (pcl3) is a covalent compound, as it is composed of nonmetals (phosphorus and chlorine), which typically form.