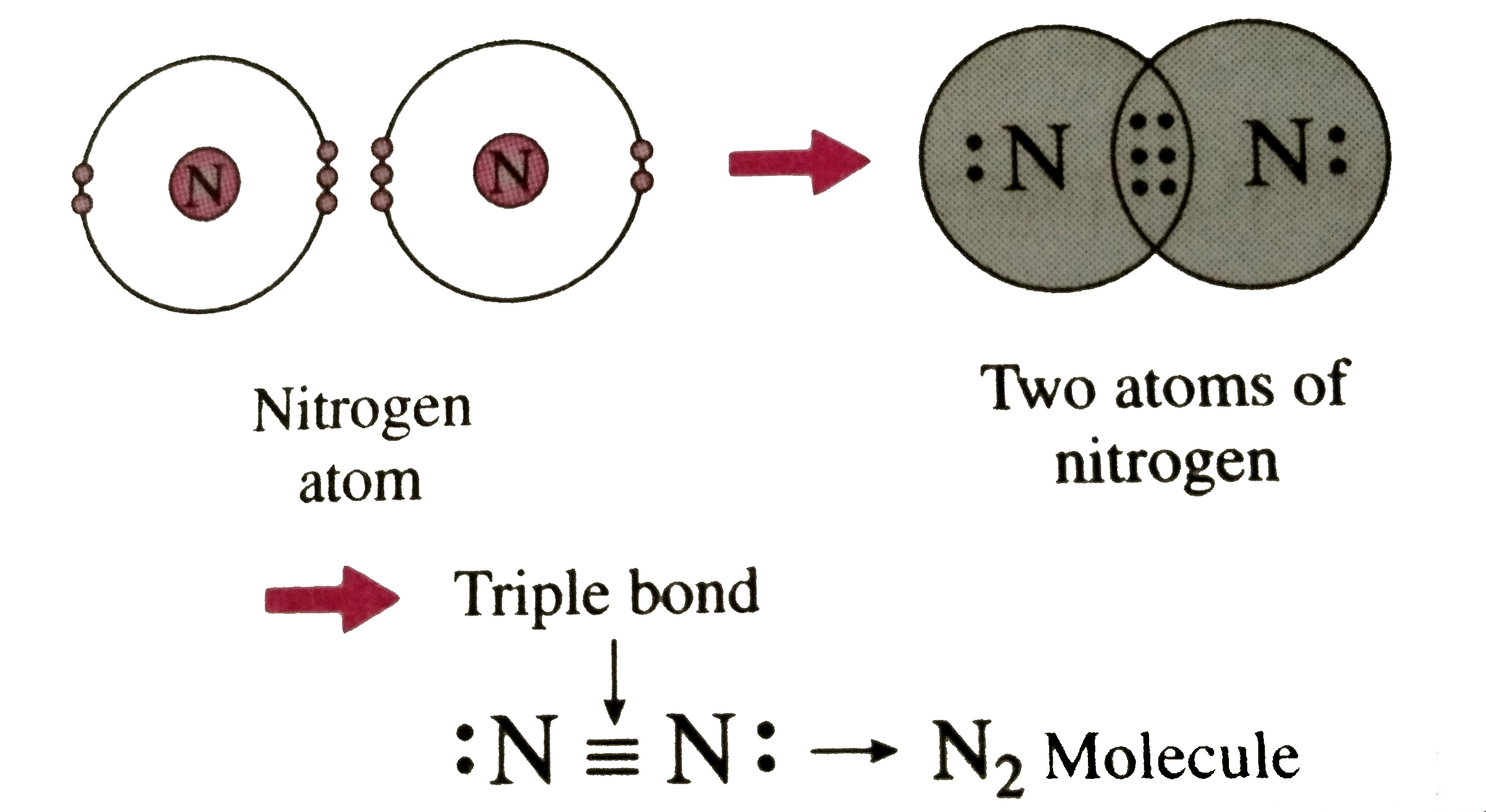How Many Bonds Can Nitrogen Form - Nitrogen is in group 5 and therefoe has 5 outer electrons. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. How many covalent bonds are in ammonia? There is a total of three covalent. Would nitrogen and phosphorus form a covalent bond? No, nitrogen and phosphorus would not typically form a covalent bond with each. Ammonia is a nitrogen atom bonded to three hydrogen atoms. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. How many covalent bonds does water have? That means it needs 3 bonds.
Nitrogen can form 3 covalent bonds and 1 coordinate bond. How many covalent bonds does water have? How many covalent bonds are in ammonia? There is a total of three covalent. Nitrogen is in group 5 and therefoe has 5 outer electrons. Ammonia is a nitrogen atom bonded to three hydrogen atoms. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. That means it needs 3 bonds. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. Would nitrogen and phosphorus form a covalent bond?
How many covalent bonds does water have? Nitrogen can form 3 covalent bonds and 1 coordinate bond. Ammonia is a nitrogen atom bonded to three hydrogen atoms. How many covalent bonds are in ammonia? As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. No, nitrogen and phosphorus would not typically form a covalent bond with each. There is a total of three covalent. Would nitrogen and phosphorus form a covalent bond? Nitrogen is in group 5 and therefoe has 5 outer electrons.
PPT Covalent Bonding and Chemical Bonds PowerPoint Presentation, free
Nitrogen is in group 5 and therefoe has 5 outer electrons. There is a total of three covalent. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. Ammonia is a nitrogen atom bonded to three hydrogen atoms. No, nitrogen and phosphorus would not typically form a covalent bond with each.
Nitrogen Covalent Bond
No, nitrogen and phosphorus would not typically form a covalent bond with each. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. Would nitrogen and phosphorus form a covalent bond? Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent.
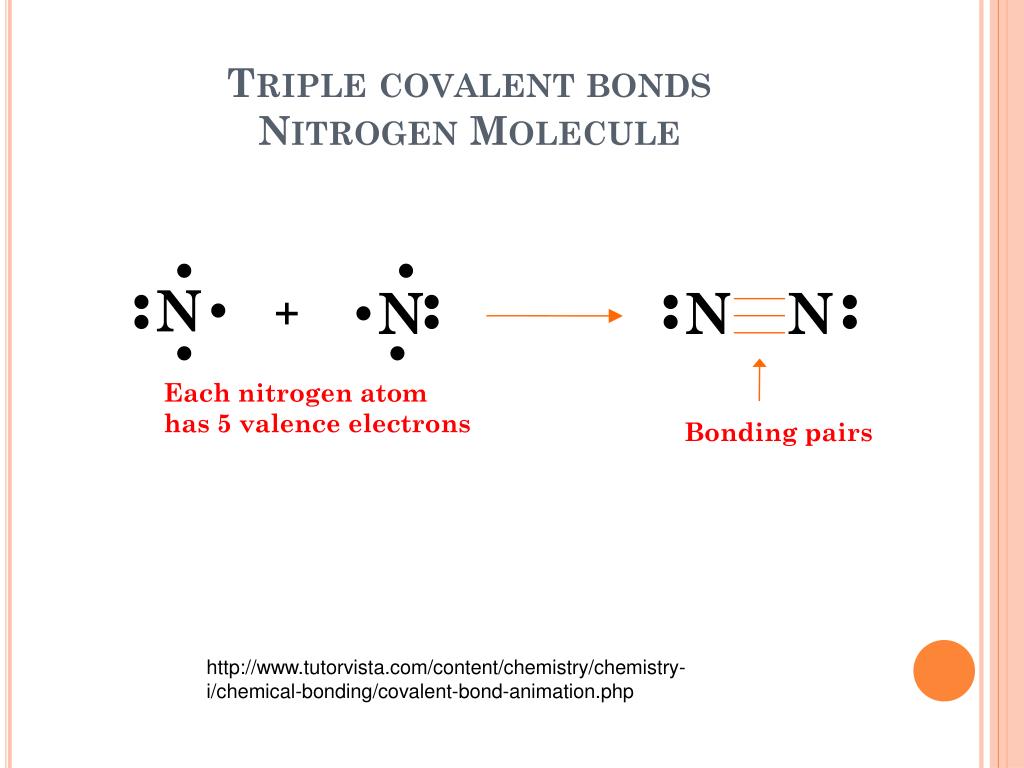
Triple Bond Nitrogen
Water molecules have two simple covalent bonds between one oxygen and two hydrogen. How many covalent bonds does water have? No, nitrogen and phosphorus would not typically form a covalent bond with each. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. That means it needs 3 bonds.
The Nitrogen Cycle. ppt download
Would nitrogen and phosphorus form a covalent bond? How many covalent bonds are in ammonia? How many covalent bonds does water have? Nitrogen can form 3 covalent bonds and 1 coordinate bond. Water molecules have two simple covalent bonds between one oxygen and two hydrogen.
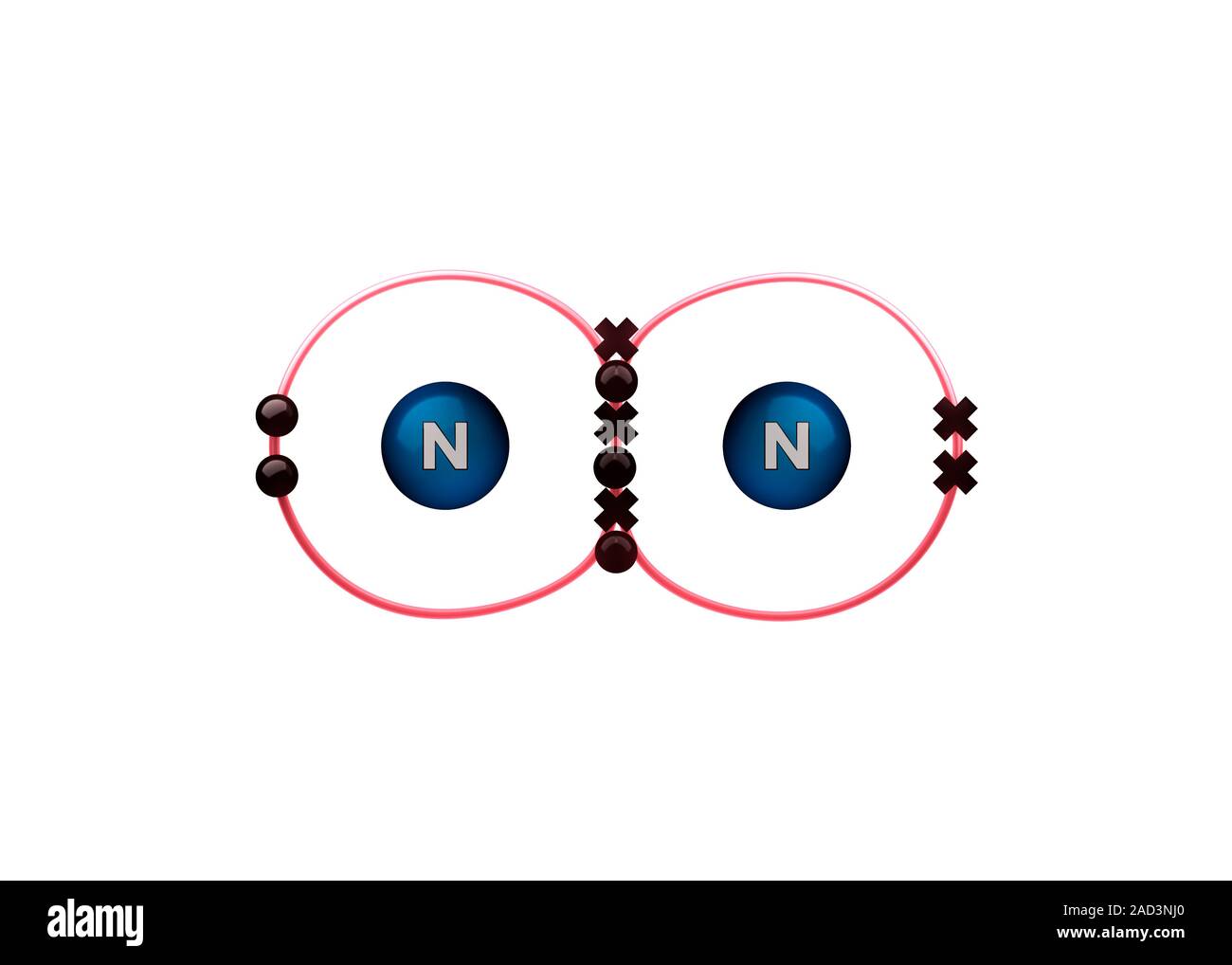
Bond formation in nitrogen molecule. Illustration of the sharing of
Ammonia is a nitrogen atom bonded to three hydrogen atoms. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. How many covalent bonds does water have? Nitrogen can form 3 covalent bonds and 1 coordinate bond.
COVALENT BONDING ORBITALS ppt download
How many covalent bonds does water have? Nitrogen can form 3 covalent bonds and 1 coordinate bond. How many covalent bonds are in ammonia? There is a total of three covalent. Would nitrogen and phosphorus form a covalent bond?
PPT Remembering General Chemistry Electronic Structure and Bonding
Would nitrogen and phosphorus form a covalent bond? There is a total of three covalent. Water molecules have two simple covalent bonds between one oxygen and two hydrogen. That means it needs 3 bonds. How many covalent bonds does water have?
Chapter 14 Ionic Bonds 2 41.49/50 = 82.98 3 37.93/50 = 75.86 ppt
No, nitrogen and phosphorus would not typically form a covalent bond with each. Would nitrogen and phosphorus form a covalent bond? That means it needs 3 bonds. How many covalent bonds are in ammonia? Nitrogen is in group 5 and therefoe has 5 outer electrons.
PPT COVALENT BONDING PowerPoint Presentation, free download ID5128236
How many covalent bonds are in ammonia? How many covalent bonds does water have? That means it needs 3 bonds. No, nitrogen and phosphorus would not typically form a covalent bond with each. Nitrogen is in group 5 and therefoe has 5 outer electrons.
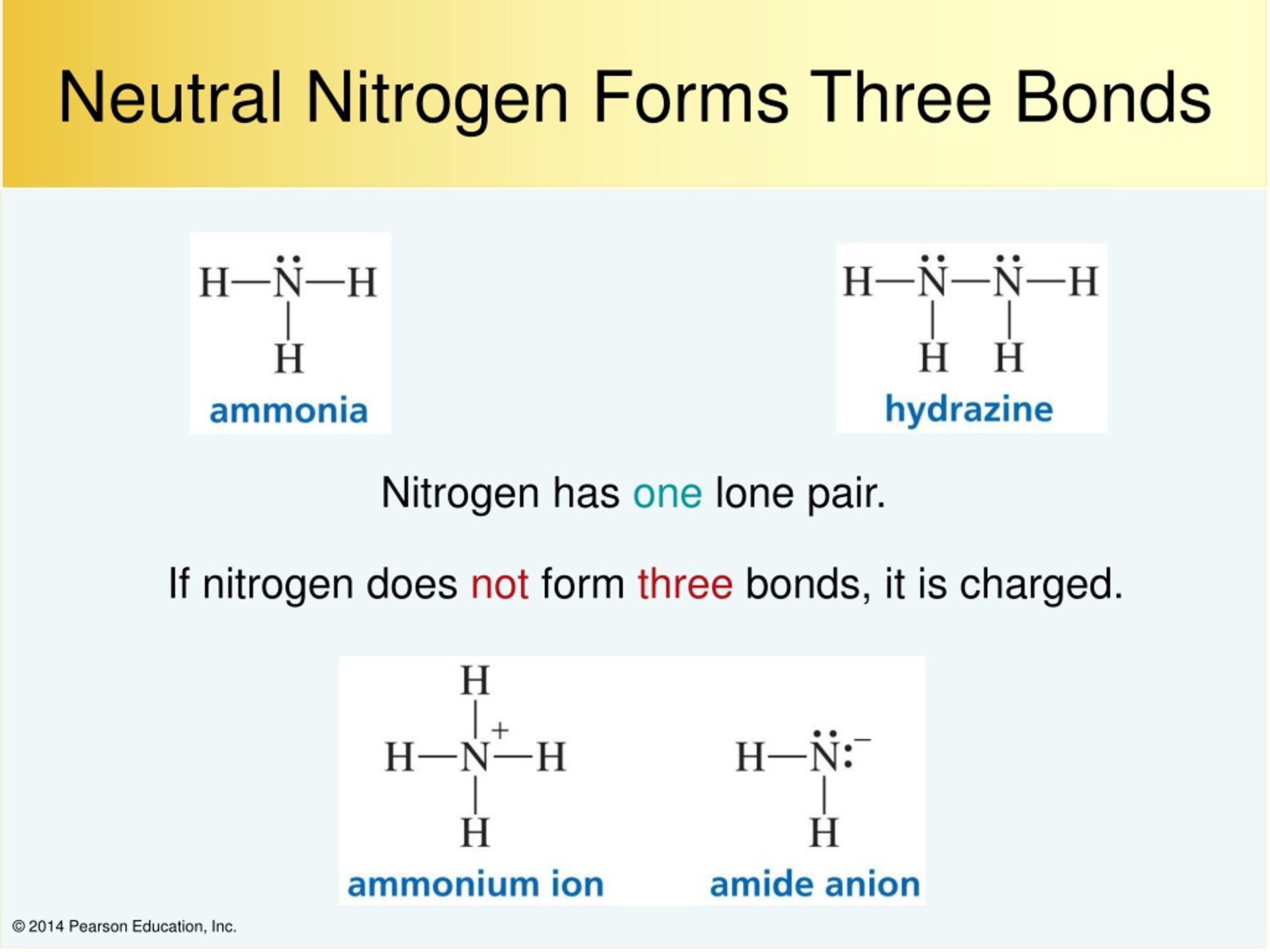
How Many Covalent Bonds Are In Ammonia?
As known, nitrogen could form 3 bonds based on octet rule, because it has 5 valence electrons. Nitrogen can form 3 covalent bonds and 1 coordinate bond. No, nitrogen and phosphorus would not typically form a covalent bond with each. How many covalent bonds does water have?
That Means It Needs 3 Bonds.
Water molecules have two simple covalent bonds between one oxygen and two hydrogen. Ammonia is a nitrogen atom bonded to three hydrogen atoms. There is a total of three covalent. Would nitrogen and phosphorus form a covalent bond?