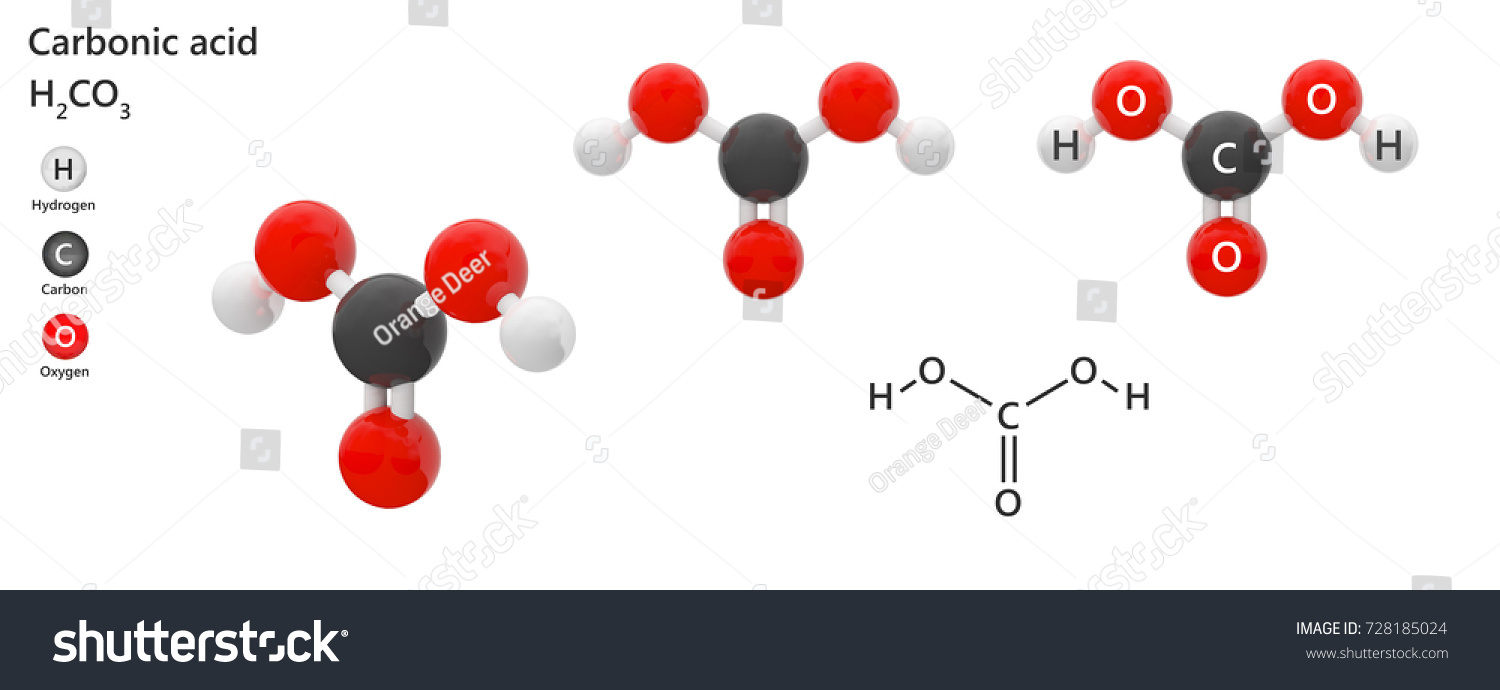
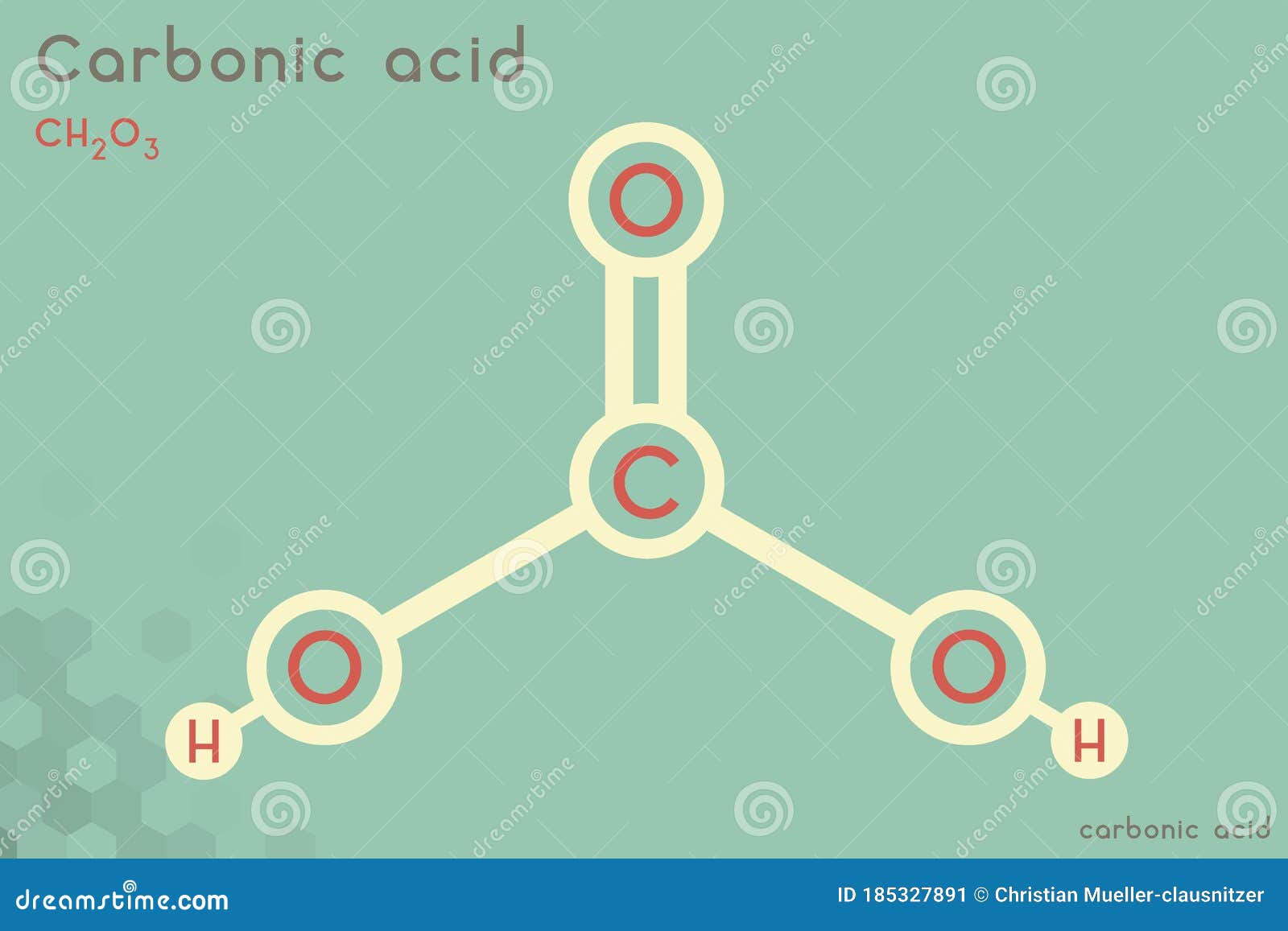
How Is Carbonic Acid Formed - In this informative video, we will uncover the fascinating process of how carbonic acid is. Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. Water and carbon dioxide combine to form carbonic acid (h 2 co 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (h +). As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions (hco 3. How is carbonic acid (h2co3) formed from carbon dioxide? Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. There are two main methods to produce anhydrous carbonic acid:
How is carbonic acid (h2co3) formed from carbon dioxide? Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions (hco 3. There are two main methods to produce anhydrous carbonic acid: In this informative video, we will uncover the fascinating process of how carbonic acid is. Water and carbon dioxide combine to form carbonic acid (h 2 co 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (h +). Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and.
As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions (hco 3. Water and carbon dioxide combine to form carbonic acid (h 2 co 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (h +). Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. How is carbonic acid (h2co3) formed from carbon dioxide? Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. In this informative video, we will uncover the fascinating process of how carbonic acid is. There are two main methods to produce anhydrous carbonic acid:
Ch 5 Weathering, soils, and erosion ppt download
There are two main methods to produce anhydrous carbonic acid: Water and carbon dioxide combine to form carbonic acid (h 2 co 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (h +). How is carbonic acid (h2co3) formed from carbon dioxide? Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. As carbon dioxide.
to Earth Science ppt download
In this informative video, we will uncover the fascinating process of how carbonic acid is. How is carbonic acid (h2co3) formed from carbon dioxide? Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its.
Carbonic Acid Chemical Compound Chemical Formula Stock Illustration
How is carbonic acid (h2co3) formed from carbon dioxide? In this informative video, we will uncover the fascinating process of how carbonic acid is. There are two main methods to produce anhydrous carbonic acid: Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂..
H2co3 Stock Vector Images Alamy
In this informative video, we will uncover the fascinating process of how carbonic acid is. Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. How is carbonic acid (h2co3) formed from carbon dioxide? As carbon dioxide enters the blood, it combines with water.
Carbonic Acid Molecule. Formed When Carbon Dioxide Is Dissolved In
Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. How is carbonic acid (h2co3) formed from carbon dioxide? Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. Water and carbon dioxide combine to form carbonic acid (h 2 co.
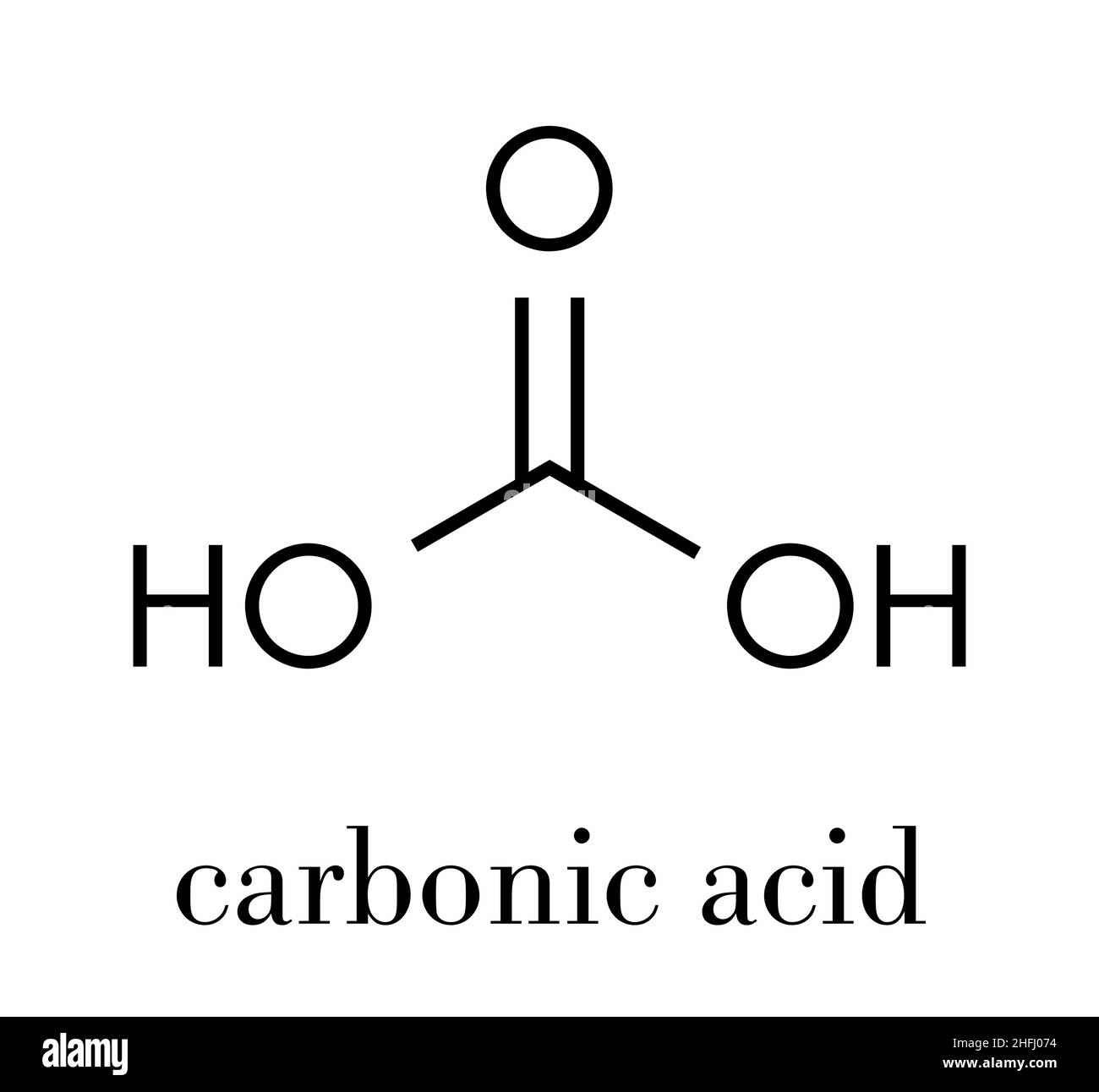
Carbonic Acid Structure
How is carbonic acid (h2co3) formed from carbon dioxide? As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions (hco 3. There are two main methods to produce anhydrous carbonic acid: Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. Formed through.
Carbonic Acid Structure
Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. Water and carbon dioxide combine to form carbonic acid (h 2 co 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (h +). As carbon dioxide enters the blood, it combines with.
How does ocean acidification change ocean chemistry? ppt download
Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and. Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. How is carbonic acid (h2co3) formed from carbon dioxide? As carbon dioxide enters the blood, it combines with water to form.
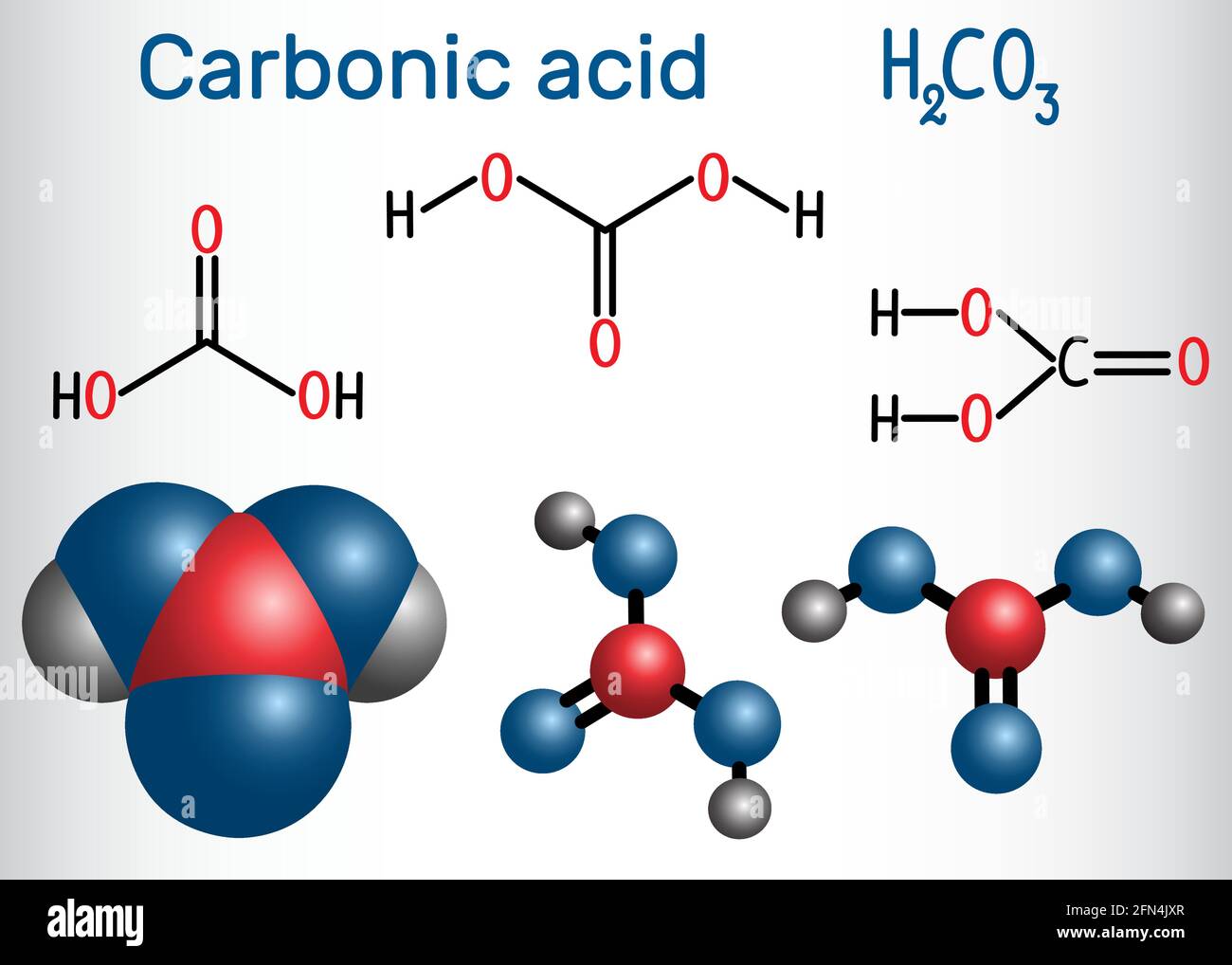
Infographic of the Molecule of Carbonic Acid Stock Vector
In this informative video, we will uncover the fascinating process of how carbonic acid is. Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. There are two main methods to produce anhydrous carbonic acid: Water and carbon dioxide combine to form carbonic acid.
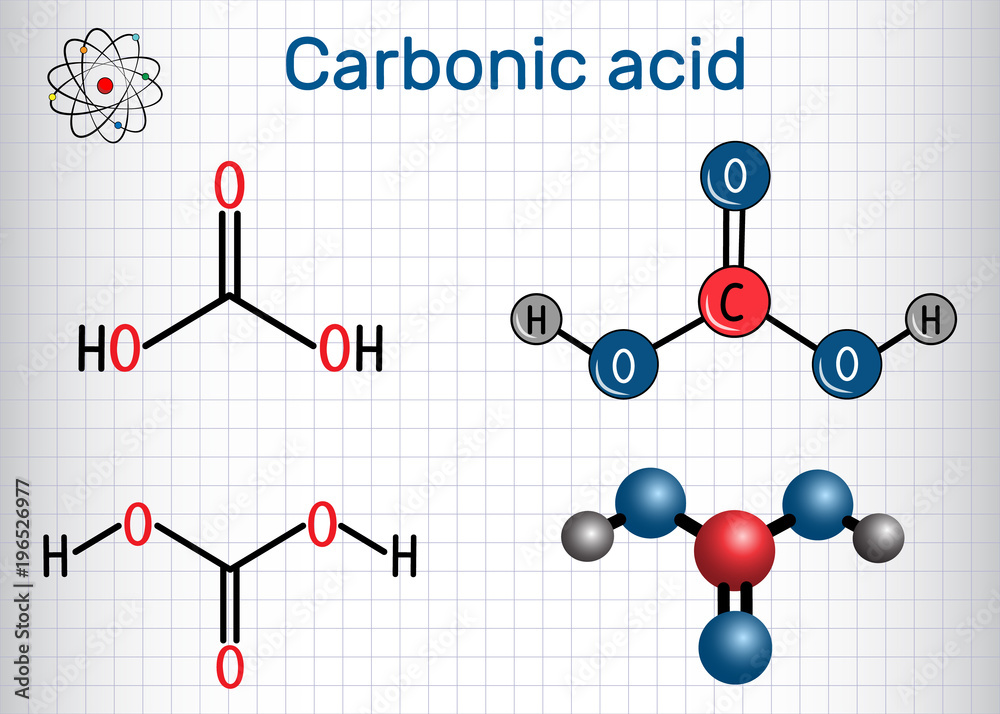
Carbonic Acid H2CO3 Molecule Structural Chemical Formula And Vector
There are two main methods to produce anhydrous carbonic acid: Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions.
Water And Carbon Dioxide Combine To Form Carbonic Acid (H 2 Co 3), A Weak Acid That Breaks (Or “Dissociates”) Into Hydrogen Ions (H +).
In this informative video, we will uncover the fascinating process of how carbonic acid is. How is carbonic acid (h2co3) formed from carbon dioxide? Formed through the reaction of carbon dioxide (co₂) with water (h₂o), carbonic acid is a weak acid that exists in equilibrium with its gaseous form, co₂. As carbon dioxide enters the blood, it combines with water to form carbonic acid, which dissociates into hydrogen ions (h +) and bicarbonate ions (hco 3.
There Are Two Main Methods To Produce Anhydrous Carbonic Acid:
Reaction of hydrogen chloride and potassium bicarbonate at 100 k in methanol and.