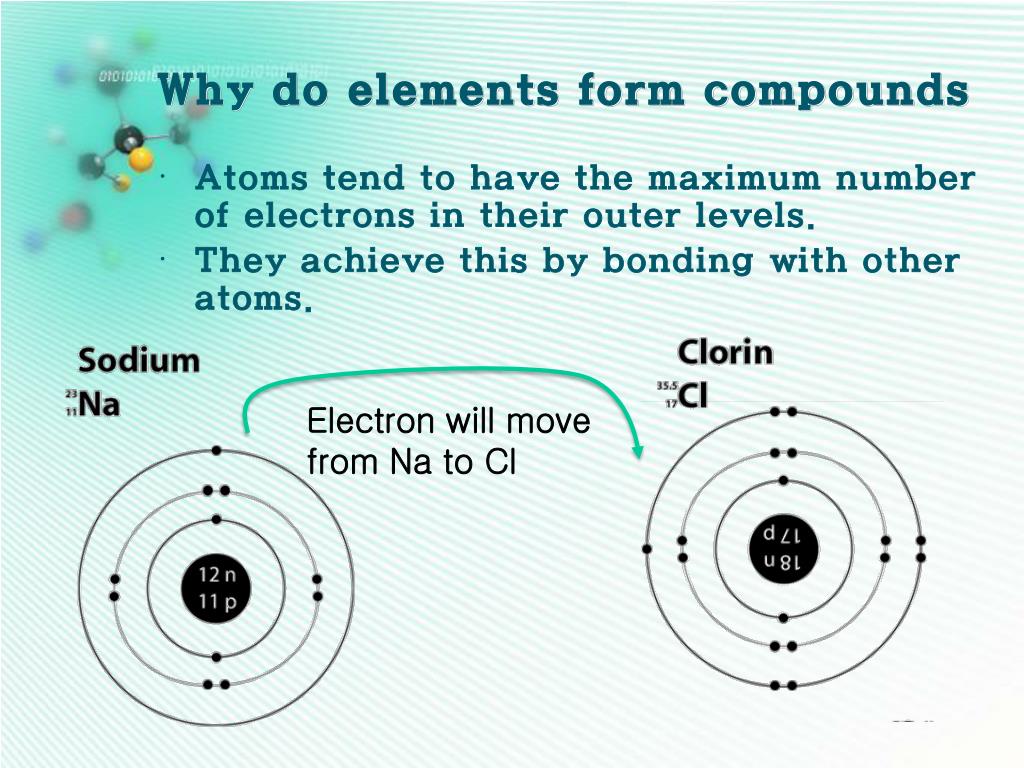
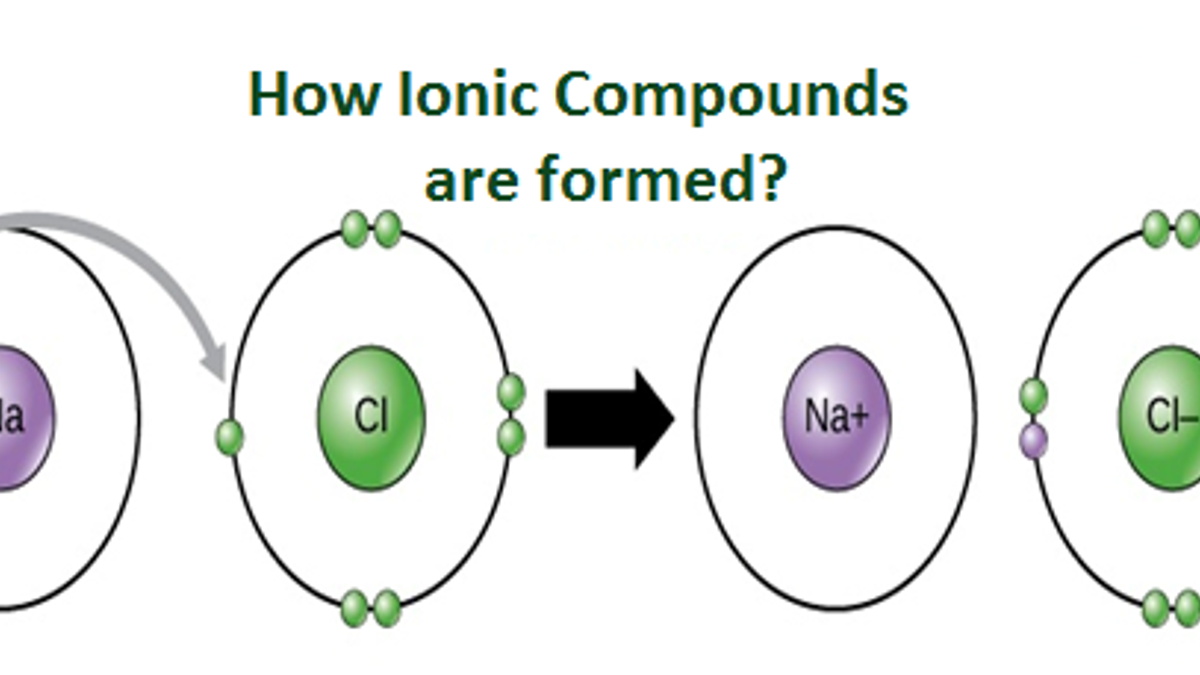
How Is A Compound Formed - The formula for the ionic compound formed between magnesium and chlorine is mgcl2. The compound formed is calcium nitrate, ca(no3)2. It is formed when one potassium ion (k+). In this compound, cesium (cs) donates one. The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. Magnesium, with a 2+ charge, forms an ion while.
The compound formed is calcium nitrate, ca(no3)2. Magnesium, with a 2+ charge, forms an ion while. The formula for the ionic compound formed between magnesium and chlorine is mgcl2. It is formed when one potassium ion (k+). In this compound, cesium (cs) donates one. The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The ionic compound formed when cs and o react is cesium oxide with the formula cs2o.
The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. The compound formed is calcium nitrate, ca(no3)2. It is formed when one potassium ion (k+). In this compound, cesium (cs) donates one. The formula for the ionic compound formed between magnesium and chlorine is mgcl2. The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. Magnesium, with a 2+ charge, forms an ion while.
Polyatomic Compounds. ppt download
The compound formed is calcium nitrate, ca(no3)2. It is formed when one potassium ion (k+). The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). Magnesium, with a 2+ charge, forms an ion while.
Chemical Bonds and forming Compounds. How is a Compound formed? A
The formula for the ionic compound formed between magnesium and chlorine is mgcl2. The compound formed is calcium nitrate, ca(no3)2. The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. In this compound, cesium (cs) donates one. The ionic compound formed when cs and o react is cesium oxide with the formula cs2o.
Chemistry The Study of Matter ppt video online download
In this compound, cesium (cs) donates one. The compound formed is calcium nitrate, ca(no3)2. The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc.
PPT Elements and compounds PowerPoint Presentation, free download
The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. It is formed when one potassium ion (k+). The formula for the ionic.
PPT Lesson Mixtures & Compounds PowerPoint Presentation, free
The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3. It is formed when one potassium ion (k+). The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). In this compound, cesium (cs) donates one. The formula for the ionic compound formed between magnesium and chlorine is mgcl2.
What is a compound? a pure substance made up of two or more types of
Magnesium, with a 2+ charge, forms an ion while. The compound formed is calcium nitrate, ca(no3)2. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). In this compound, cesium (cs) donates one. The formula for the ionic compound formed between magnesium and chlorine is mgcl2.
PPT Unit 7 Section 1 Notes PowerPoint Presentation, free download
The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). It is formed when one potassium ion (k+). The compound formed is calcium nitrate, ca(no3)2. The formula for the ionic compound formed between magnesium and chlorine is mgcl2.
Compound Turbo 12 Valve
The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). The compound formed is calcium nitrate, ca(no3)2. In this compound, cesium (cs) donates one. It is formed when one potassium ion (k+). The formula for the ionic compound formed between magnesium and chlorine is mgcl2.
Properties of Ionic and molecular compounds ppt download
The compound formed is calcium nitrate, ca(no3)2. The formula for the ionic compound formed between magnesium and chlorine is mgcl2. It is formed when one potassium ion (k+). The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). Magnesium, with a 2+ charge, forms an ion while.
What are Ionic Compounds and how they are formed?
The compound formed from the ions of potassium and chlorine is potassium chloride (kcl). In this compound, cesium (cs) donates one. The ionic compound formed when cs and o react is cesium oxide with the formula cs2o. Magnesium, with a 2+ charge, forms an ion while. The compound formed is calcium nitrate, ca(no3)2.
It Is Formed When One Potassium Ion (K+).
Magnesium, with a 2+ charge, forms an ion while. The formula for the ionic compound formed between magnesium and chlorine is mgcl2. In this compound, cesium (cs) donates one. The compound formed from the ions of potassium and chlorine is potassium chloride (kcl).
The Ionic Compound Formed When Cs And O React Is Cesium Oxide With The Formula Cs2O.
The compound formed is calcium nitrate, ca(no3)2. The chemical formula for the compound formed between scandium (iii) and the bromate ion is sc (bro3)3.