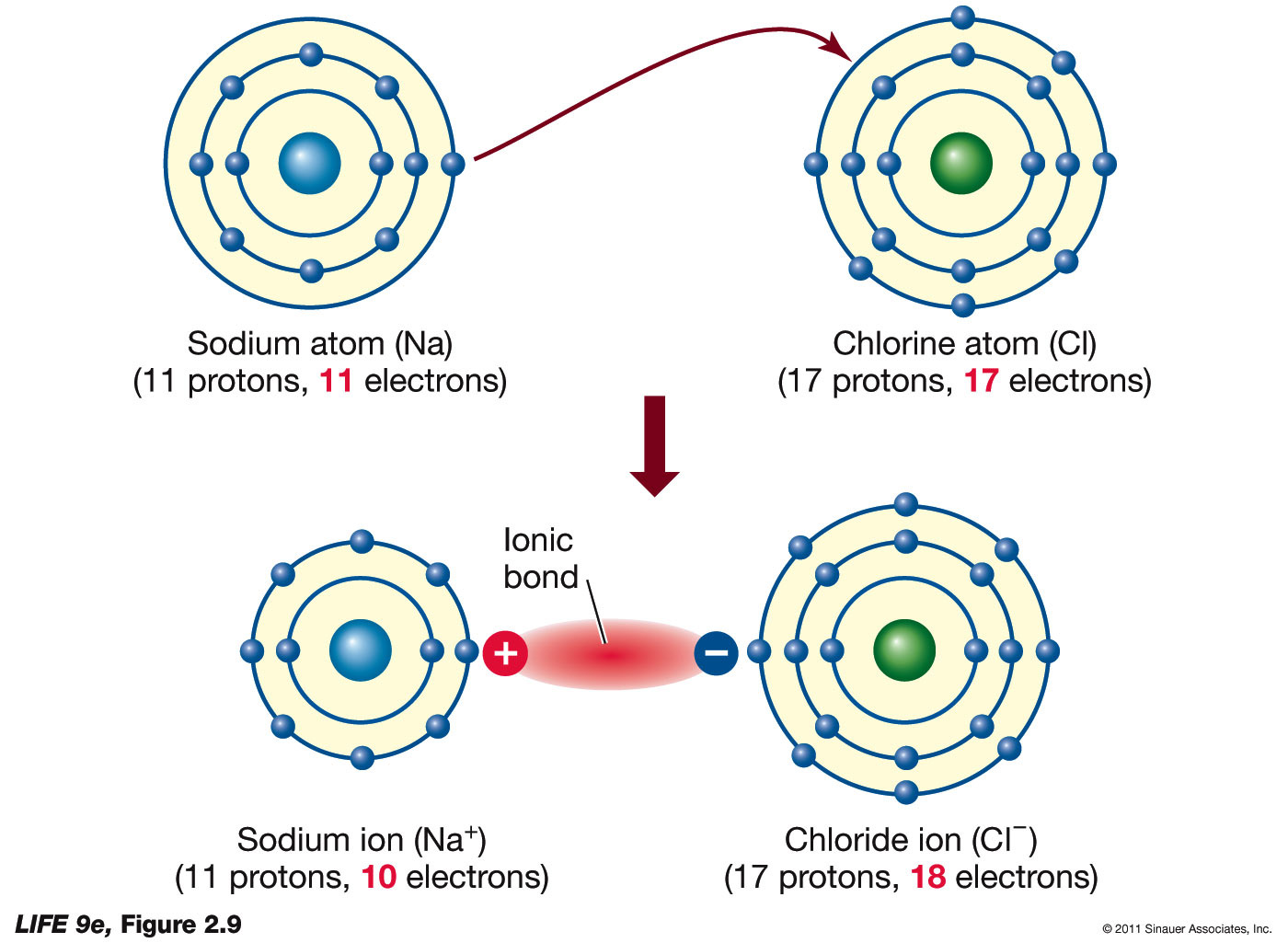
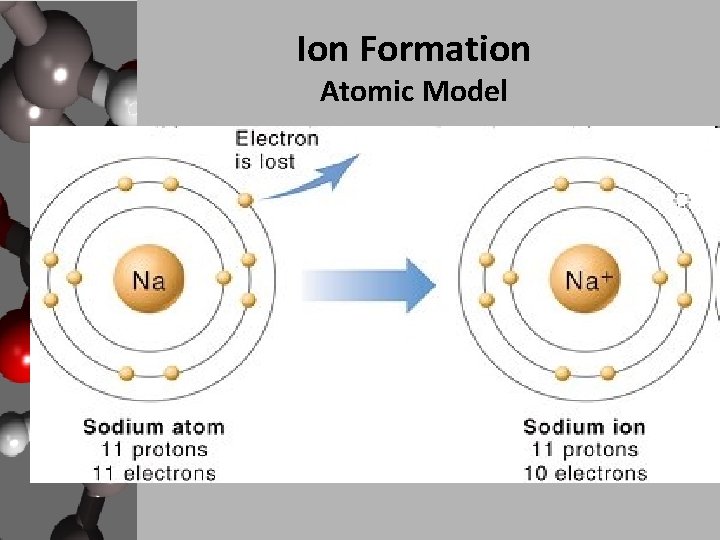
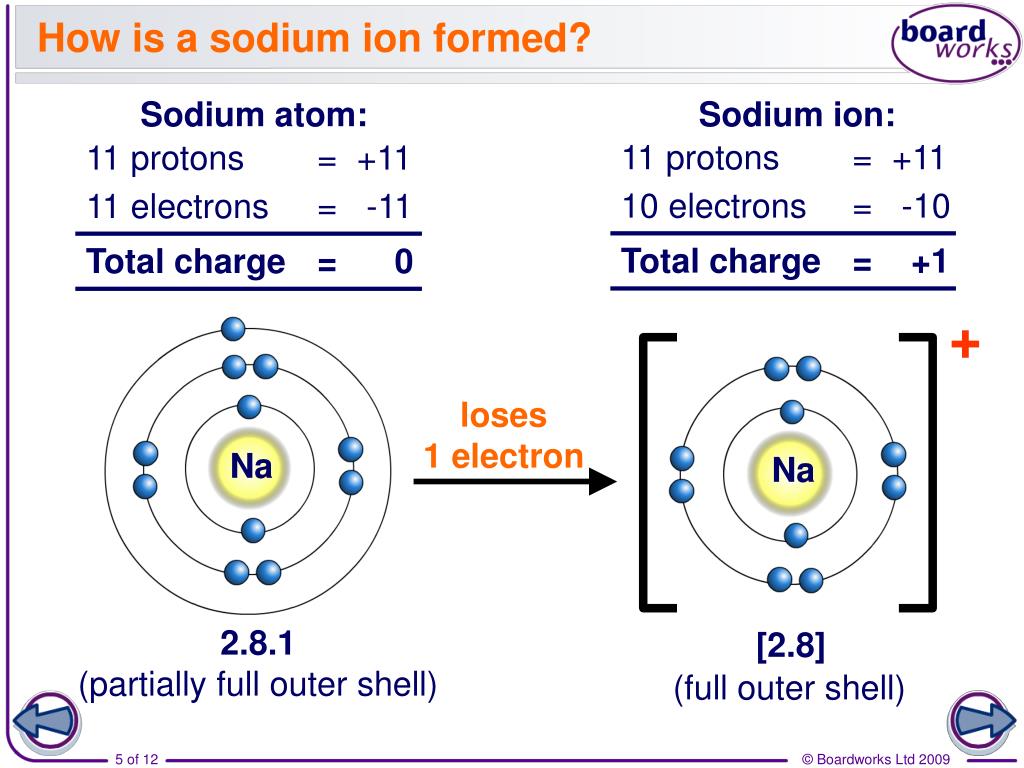
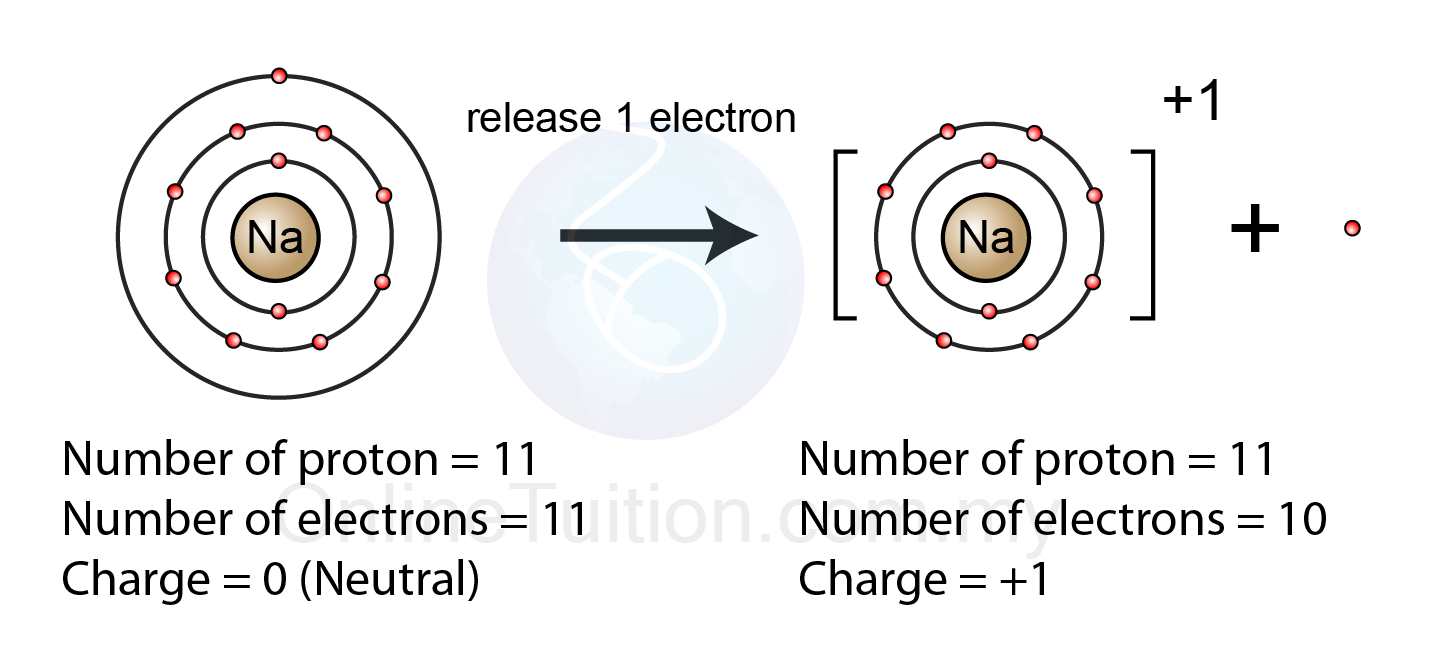
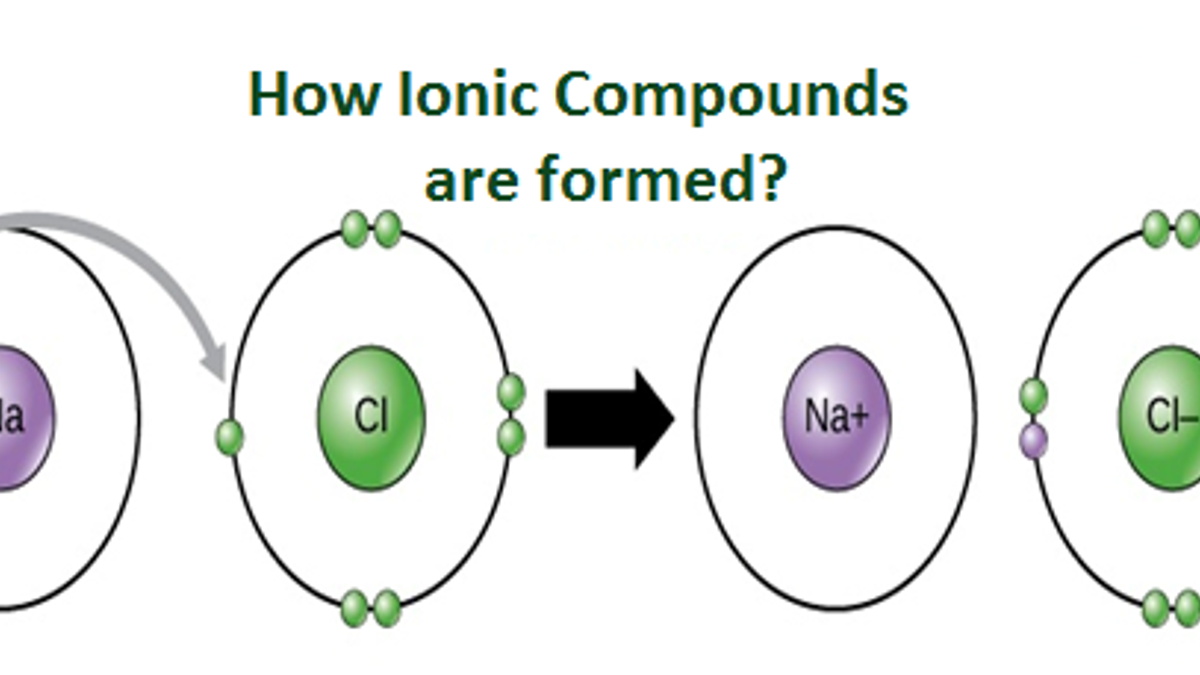
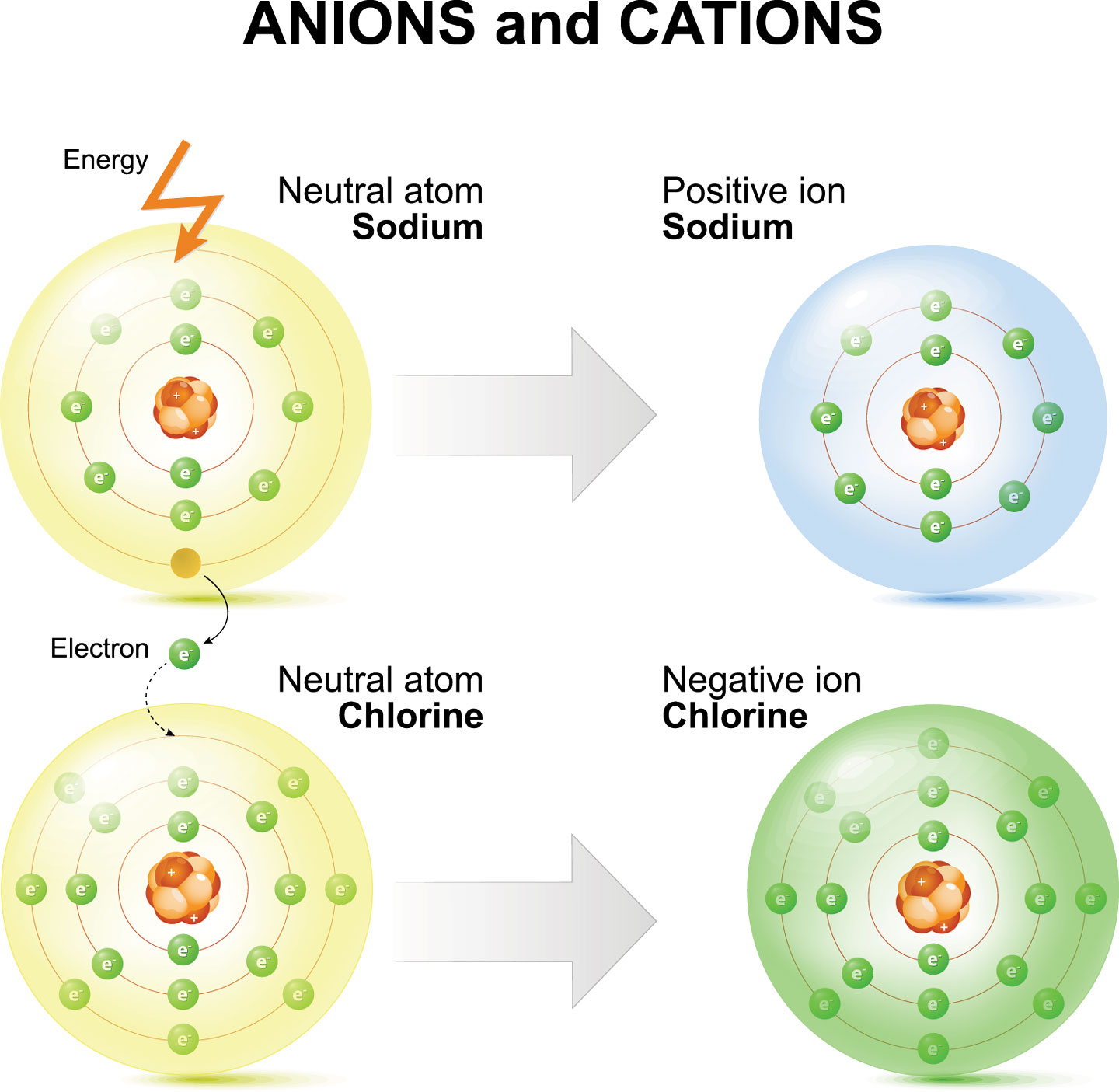
How Does An Ion Form - This stable electron configuration prevents argon from. However, there are multiple lead cations, each with a different charge. Argon typically does not form ions because it has a full outer electron shell. No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. The lead ion is a cation, meaning that it has a positive charge. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+. Does silicon form an ion? Potassium has to lose 1 electron in order to form an ion. Once it does, it is known as a cation (ion with a positive charge,) and its symbol.
No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+. Potassium has to lose 1 electron in order to form an ion. The lead ion is a cation, meaning that it has a positive charge. Argon typically does not form ions because it has a full outer electron shell. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. Does silicon form an ion? However, there are multiple lead cations, each with a different charge. This stable electron configuration prevents argon from.
Argon typically does not form ions because it has a full outer electron shell. This stable electron configuration prevents argon from. However, there are multiple lead cations, each with a different charge. Does silicon form an ion? No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. Potassium has to lose 1 electron in order to form an ion. The lead ion is a cation, meaning that it has a positive charge. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+.
Diagram Of An Ion Ionic Compound Bond Examples Bonding Examp
Potassium has to lose 1 electron in order to form an ion. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. This stable electron configuration prevents argon from. Does silicon form an ion? No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than.
Ionic Compounds and Metals Bonding and Properties Chapter
This stable electron configuration prevents argon from. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. Does silicon form an ion? Argon typically does not form ions because it has a full outer electron shell. However, there are multiple lead cations, each with a different charge.
PPT How do atoms form ions? PowerPoint Presentation, free download
Once it does, it is known as a cation (ion with a positive charge,) and its symbol. However, there are multiple lead cations, each with a different charge. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+. No, silicon generally does not form ions because.
Ionic Bonds Introduction to Chemistry Chapter 4 Lesson 2 ppt download
Potassium has to lose 1 electron in order to form an ion. Argon typically does not form ions because it has a full outer electron shell. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. The lead ion is a cation, meaning that it has a positive charge. However, there are multiple.
Ionic Bond Formation Edexcel GCSE Chemistry Revision
Does silicon form an ion? However, there are multiple lead cations, each with a different charge. The lead ion is a cation, meaning that it has a positive charge. This stable electron configuration prevents argon from. Argon typically does not form ions because it has a full outer electron shell.
Formation of Ion SPM Chemistry
However, there are multiple lead cations, each with a different charge. No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. Does silicon form an ion? However, h+ will not stay a free ion, and.
Ionic Bonds Introduction to Chemistry Chapter 4 Lesson 2 ppt download
No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. Potassium has to lose 1 electron in order to form an ion. However, there are multiple lead cations, each with a different charge. This stable.
What are Ionic Compounds and how they are formed?
Does silicon form an ion? Argon typically does not form ions because it has a full outer electron shell. However, there are multiple lead cations, each with a different charge. Potassium has to lose 1 electron in order to form an ion. Once it does, it is known as a cation (ion with a positive charge,) and its symbol.
Ion Definition and Examples Biology Online Dictionary
This stable electron configuration prevents argon from. The lead ion is a cation, meaning that it has a positive charge. However, there are multiple lead cations, each with a different charge. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. However, h+ will not stay a free ion, and instead immediately combines.
Explainer Ions and radicals in our world
Potassium has to lose 1 electron in order to form an ion. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+. This stable electron configuration prevents argon from. Once it does, it is known as a cation (ion with a positive charge,) and its symbol..
Does Silicon Form An Ion?
However, there are multiple lead cations, each with a different charge. This stable electron configuration prevents argon from. The lead ion is a cation, meaning that it has a positive charge. Argon typically does not form ions because it has a full outer electron shell.
Potassium Has To Lose 1 Electron In Order To Form An Ion.
No, silicon generally does not form ions because it tends to share electrons in covalent bonds rather than. Once it does, it is known as a cation (ion with a positive charge,) and its symbol. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule (h2o) to form the hydronium ion, h3o+.