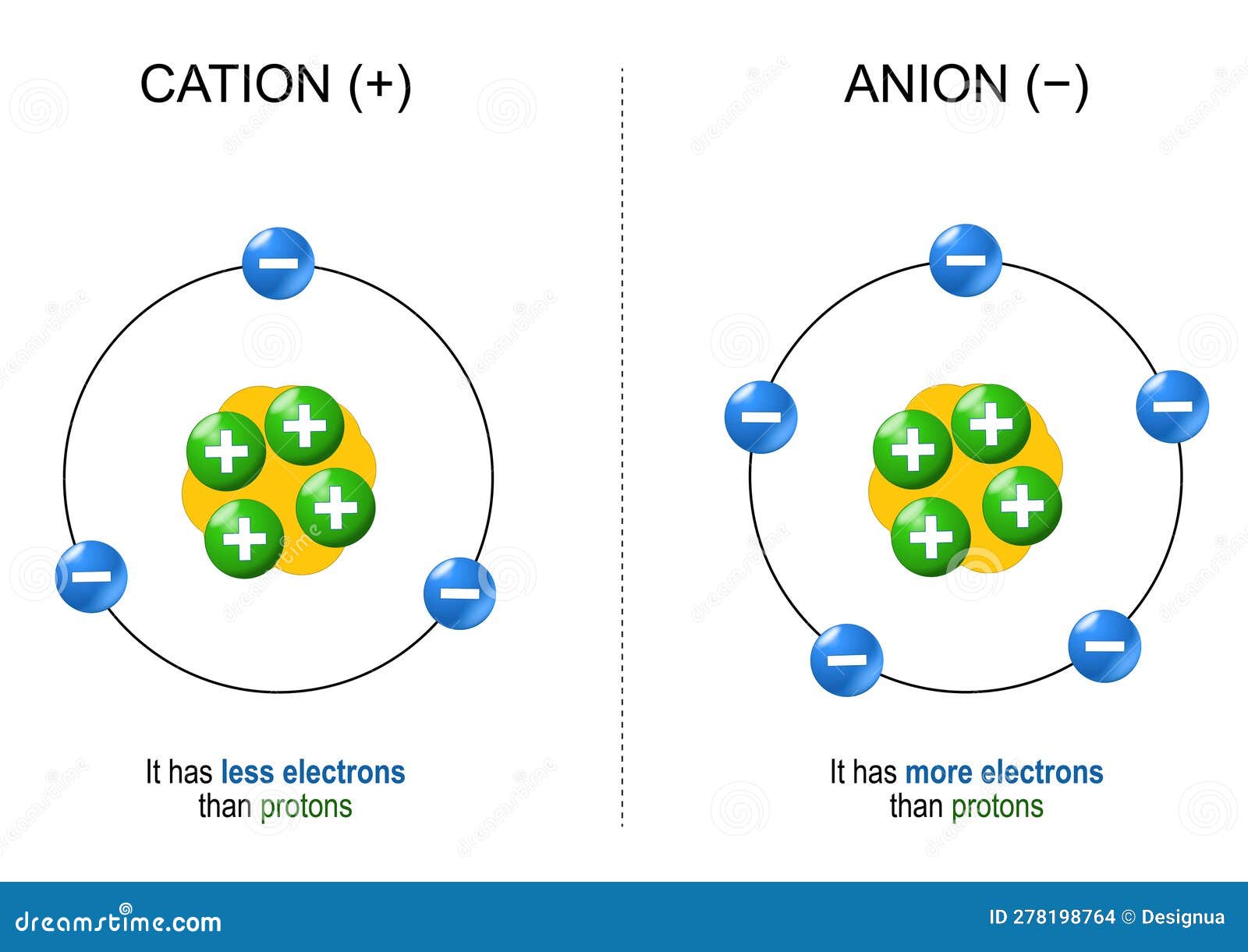
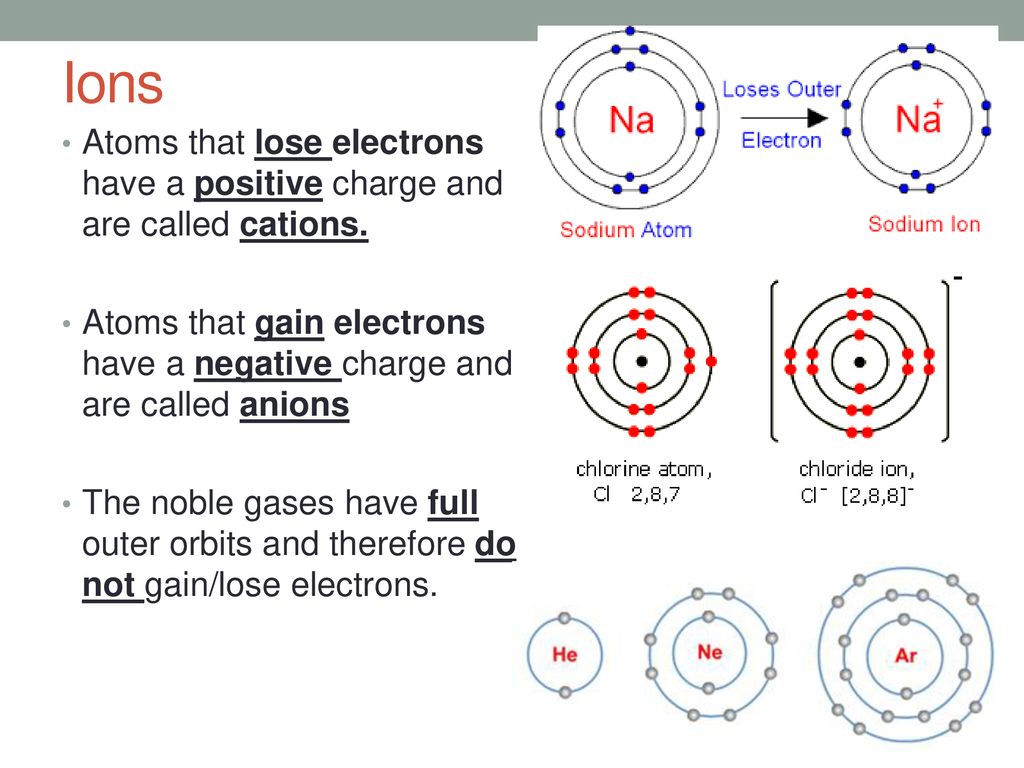
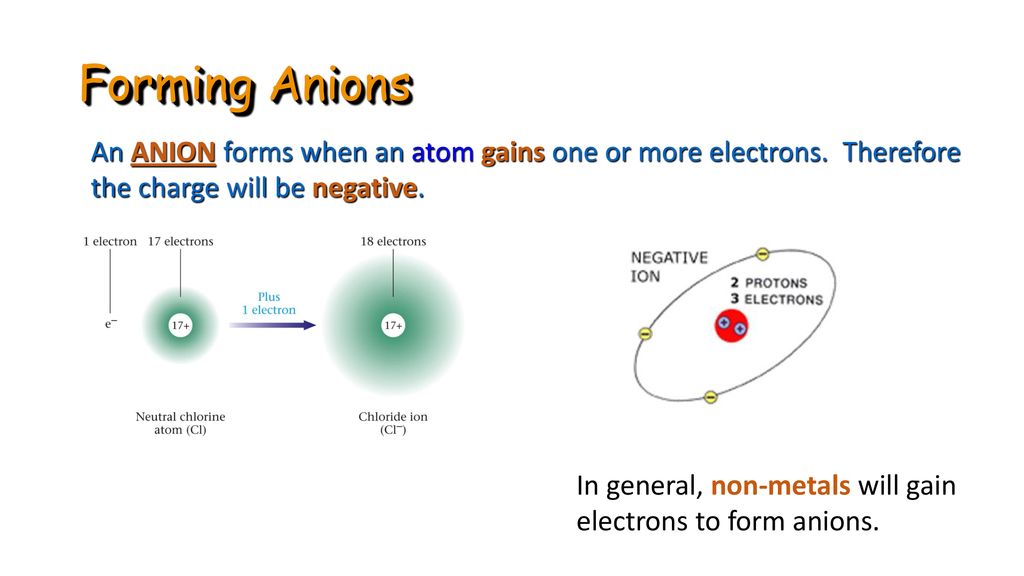
How Does An Anion Form - This stable electron configuration prevents argon from. Borides which could be thought to contain anions are covalent. Boron does not form cations or anions in compounds. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. An iodine is an anion, or a negatively charged ion. What type of ion does nitrogen form? Argon typically does not form ions because it has a full outer electron shell. Scandium is a cation because it tends to lose.
Borides which could be thought to contain anions are covalent. Argon typically does not form ions because it has a full outer electron shell. What type of ion does nitrogen form? Boron does not form cations or anions in compounds. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. An iodine is an anion, or a negatively charged ion. This stable electron configuration prevents argon from. Scandium is a cation because it tends to lose. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable.
This stable electron configuration prevents argon from. An iodine is an anion, or a negatively charged ion. Boron does not form cations or anions in compounds. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. Scandium is a cation because it tends to lose. What type of ion does nitrogen form? Borides which could be thought to contain anions are covalent. Argon typically does not form ions because it has a full outer electron shell.
Cations and Anions. Structure of Ions Stock Vector Illustration of
Boron does not form cations or anions in compounds. Argon typically does not form ions because it has a full outer electron shell. An iodine is an anion, or a negatively charged ion. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. What type of ion does nitrogen form?
Objectives To describe the formation of ions from their parent atoms
An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. This stable electron configuration prevents argon from. Argon typically does not form ions because it has a full outer electron shell. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. An iodine is an anion, or.
Ions and Ionic Compounds ppt download
Argon typically does not form ions because it has a full outer electron shell. Boron does not form cations or anions in compounds. Scandium is a cation because it tends to lose. This stable electron configuration prevents argon from. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable.
Ions. ppt download
Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. An iodine is an anion, or a negatively charged ion. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. Scandium is a cation because it tends to lose. What type of ion does nitrogen form?
Cations vs Anions
An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. An iodine is an anion, or a negatively charged ion. Scandium is a cation because it tends to lose. This stable electron configuration prevents argon from. Boron does not form cations or anions in compounds.
Chapter 6 Bonding. ppt download
Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. Borides which could be thought to contain anions are covalent. Boron does not form cations or anions in compounds. What type of ion does nitrogen form? An iodine is an anion, or a negatively charged ion.
PPT IONS PowerPoint Presentation, free download ID2435906
An iodine is an anion, or a negatively charged ion. Scandium is a cation because it tends to lose. What type of ion does nitrogen form? Argon typically does not form ions because it has a full outer electron shell. Borides which could be thought to contain anions are covalent.
Structure & Bonding. ppt download
Boron does not form cations or anions in compounds. An iodine is an anion, or a negatively charged ion. Argon typically does not form ions because it has a full outer electron shell. Scandium is a cation because it tends to lose. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding.
Anion Atom
An iodine is an anion, or a negatively charged ion. Boron does not form cations or anions in compounds. Scandium is a cation because it tends to lose. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. Argon typically does not form ions because it has a full outer electron shell.
Cations vs Anions Difference Between Cations and Anions with Examples
Scandium is a cation because it tends to lose. Boron does not form cations or anions in compounds. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. An iodine is an anion, or a negatively charged.
Boron Does Not Form Cations Or Anions In Compounds.
An anion is formed from an atom by the gain of electrons, a process known as ionic bonding. Nitrogen typically forms a negative ion (anion) by gaining three electrons to achieve a stable. An iodine is an anion, or a negatively charged ion. What type of ion does nitrogen form?
Argon Typically Does Not Form Ions Because It Has A Full Outer Electron Shell.
Borides which could be thought to contain anions are covalent. This stable electron configuration prevents argon from. Scandium is a cation because it tends to lose.