How Does A Cation Form - The lead ion is a cation, meaning that it has a positive charge. Scandium is a cation because it tends to lose electrons to form a positive charge. In chemical compounds, boron tends to form covalent bonds. Is boron a cation or an anion? Boron is a metalloid element with the symbol b. However, there are multiple lead cations, each with a different charge. This stable electron configuration prevents argon from. Fluorine does not form cations, or any compound, complex ion, or coordinate. Argon typically does not form ions because it has a full outer electron shell. Fluorine is a neutral atom, though fluoride is an anion.
Is boron a cation or an anion? Fluorine is a neutral atom, though fluoride is an anion. Scandium is a cation because it tends to lose electrons to form a positive charge. In chemical compounds, boron tends to form covalent bonds. Argon typically does not form ions because it has a full outer electron shell. This stable electron configuration prevents argon from. Boron is a metalloid element with the symbol b. The lead ion is a cation, meaning that it has a positive charge. Fluorine does not form cations, or any compound, complex ion, or coordinate. However, there are multiple lead cations, each with a different charge.
This stable electron configuration prevents argon from. Is boron a cation or an anion? Scandium is a cation because it tends to lose electrons to form a positive charge. Argon typically does not form ions because it has a full outer electron shell. Fluorine does not form cations, or any compound, complex ion, or coordinate. However, there are multiple lead cations, each with a different charge. The lead ion is a cation, meaning that it has a positive charge. Boron is a metalloid element with the symbol b. Fluorine is a neutral atom, though fluoride is an anion. In chemical compounds, boron tends to form covalent bonds.
Explainer Ions and radicals in our world Science News for Students
Fluorine is a neutral atom, though fluoride is an anion. Fluorine does not form cations, or any compound, complex ion, or coordinate. However, there are multiple lead cations, each with a different charge. In chemical compounds, boron tends to form covalent bonds. Argon typically does not form ions because it has a full outer electron shell.
Valence elecrtrons and chemical properties ppt download
Fluorine does not form cations, or any compound, complex ion, or coordinate. Argon typically does not form ions because it has a full outer electron shell. Is boron a cation or an anion? This stable electron configuration prevents argon from. The lead ion is a cation, meaning that it has a positive charge.
Chapters 7 and 8 Bonding. ppt download
Is boron a cation or an anion? Scandium is a cation because it tends to lose electrons to form a positive charge. Fluorine is a neutral atom, though fluoride is an anion. This stable electron configuration prevents argon from. Fluorine does not form cations, or any compound, complex ion, or coordinate.
Unit 5 Ionic Bonding & Nomenclature ppt download
However, there are multiple lead cations, each with a different charge. Is boron a cation or an anion? Argon typically does not form ions because it has a full outer electron shell. The lead ion is a cation, meaning that it has a positive charge. Scandium is a cation because it tends to lose electrons to form a positive charge.
Chapter 6B Chemical Bonding ppt download
In chemical compounds, boron tends to form covalent bonds. Scandium is a cation because it tends to lose electrons to form a positive charge. Argon typically does not form ions because it has a full outer electron shell. Fluorine does not form cations, or any compound, complex ion, or coordinate. Fluorine is a neutral atom, though fluoride is an anion.
Chapter 6 Bonding. ppt download
In chemical compounds, boron tends to form covalent bonds. Argon typically does not form ions because it has a full outer electron shell. Fluorine does not form cations, or any compound, complex ion, or coordinate. Scandium is a cation because it tends to lose electrons to form a positive charge. Fluorine is a neutral atom, though fluoride is an anion.
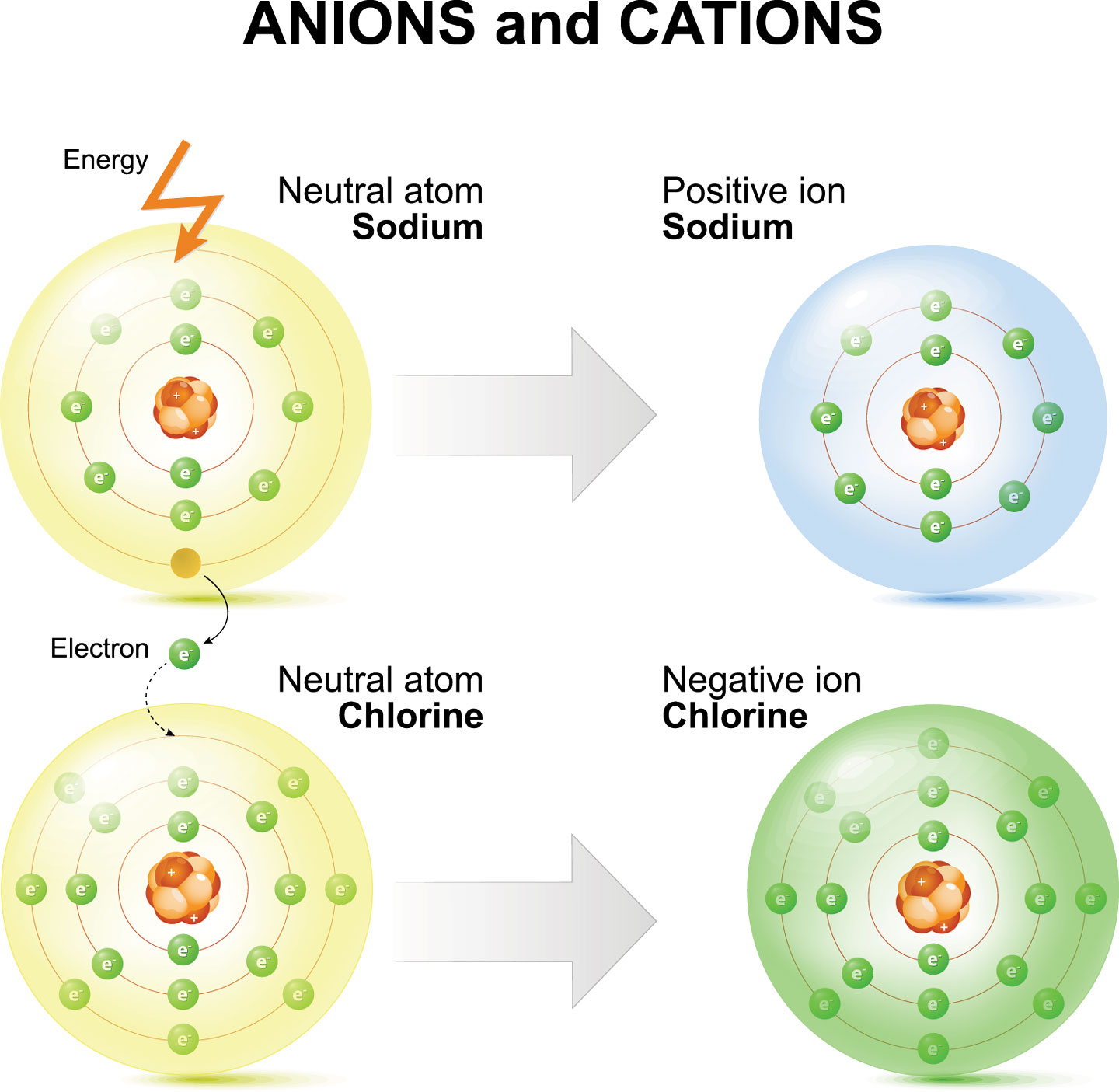
Cations and Anions Definitions, Examples, and Differences
However, there are multiple lead cations, each with a different charge. Argon typically does not form ions because it has a full outer electron shell. The lead ion is a cation, meaning that it has a positive charge. Fluorine is a neutral atom, though fluoride is an anion. Scandium is a cation because it tends to lose electrons to form.
Structure & Bonding. ppt download
In chemical compounds, boron tends to form covalent bonds. Argon typically does not form ions because it has a full outer electron shell. However, there are multiple lead cations, each with a different charge. Fluorine is a neutral atom, though fluoride is an anion. The lead ion is a cation, meaning that it has a positive charge.
Ions. ppt download
Boron is a metalloid element with the symbol b. Is boron a cation or an anion? This stable electron configuration prevents argon from. However, there are multiple lead cations, each with a different charge. In chemical compounds, boron tends to form covalent bonds.
Argon Typically Does Not Form Ions Because It Has A Full Outer Electron Shell.
Boron is a metalloid element with the symbol b. In chemical compounds, boron tends to form covalent bonds. Fluorine is a neutral atom, though fluoride is an anion. This stable electron configuration prevents argon from.
Fluorine Does Not Form Cations, Or Any Compound, Complex Ion, Or Coordinate.
Scandium is a cation because it tends to lose electrons to form a positive charge. However, there are multiple lead cations, each with a different charge. The lead ion is a cation, meaning that it has a positive charge. Is boron a cation or an anion?



:+This+loss+of+electrons+is+called+oxidation..jpg)




