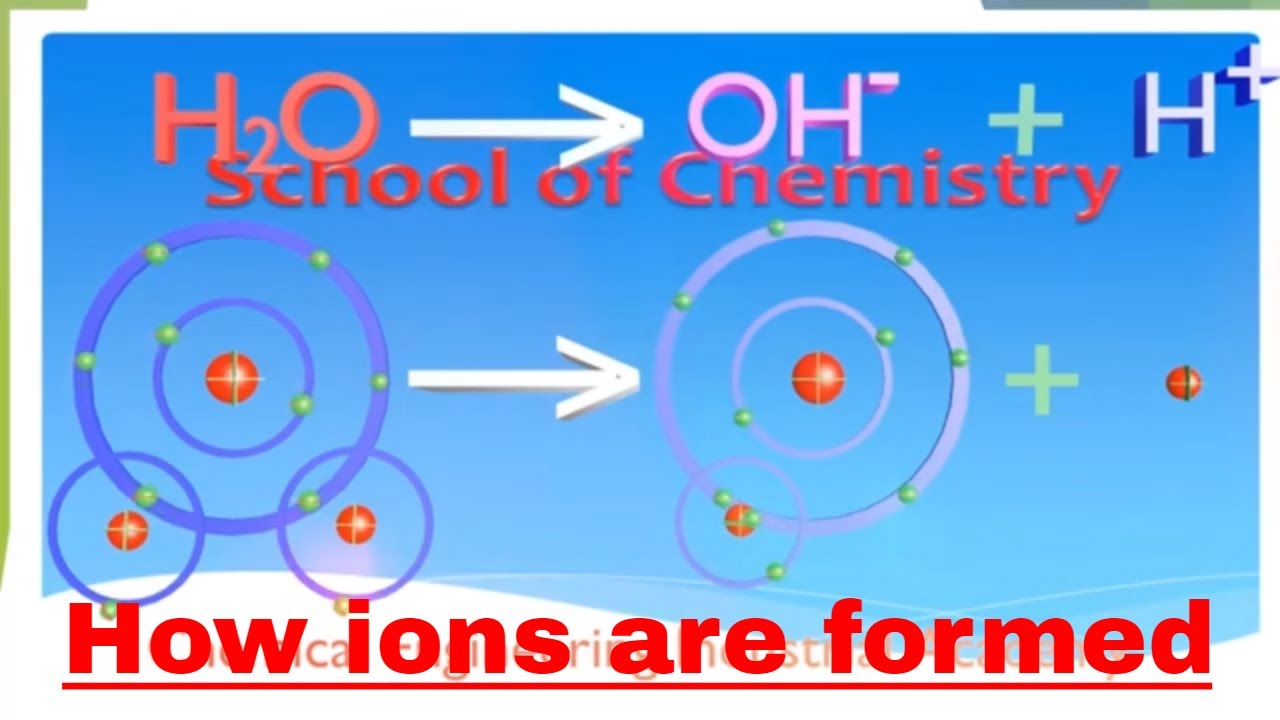
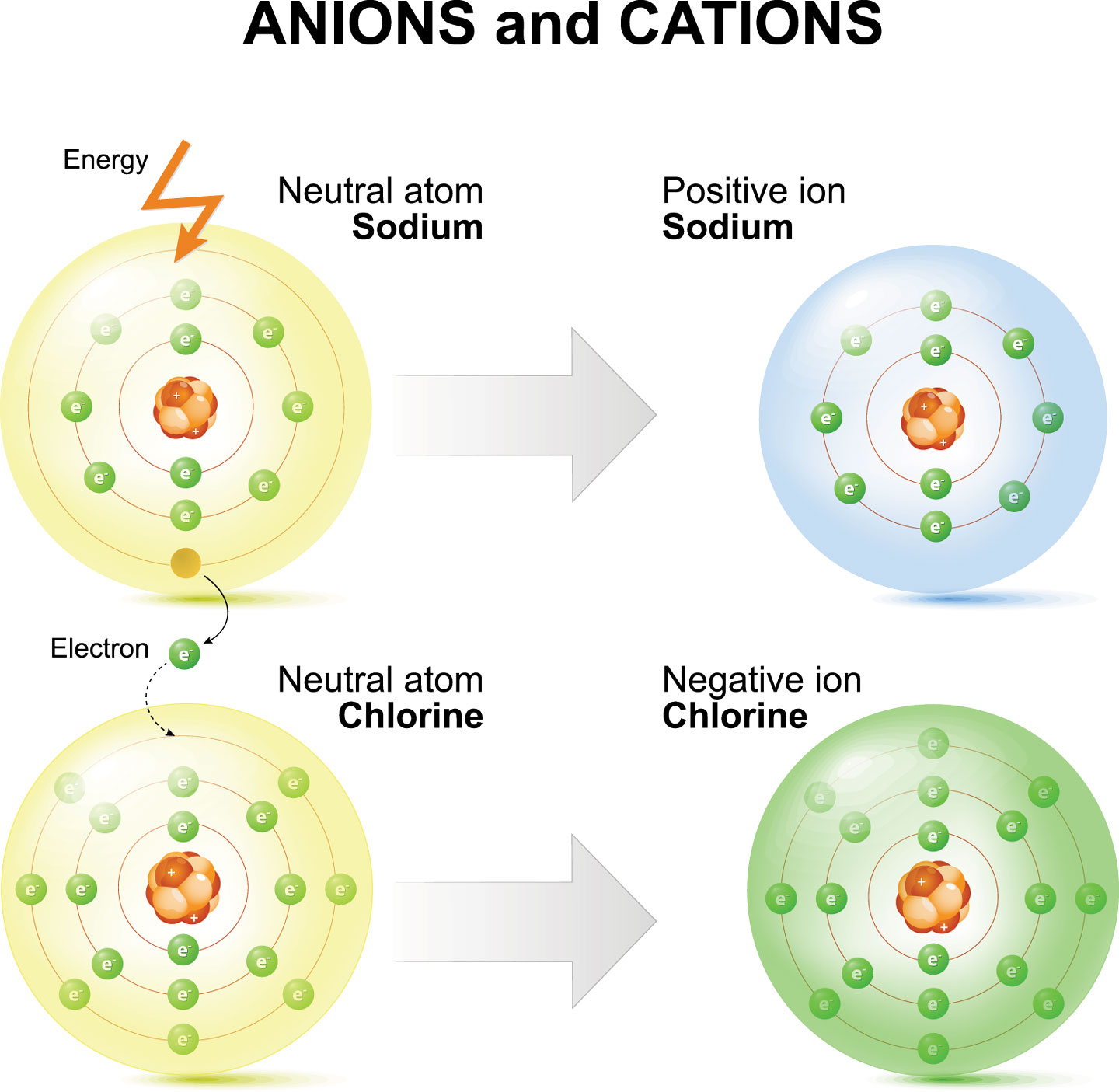
How Are Ions Formed - Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. Sodium easily loses one electron to achieve a. Sulfuric acid is a strong acid that typically dissociates into hydronium ions and. Ions formed from sodium atoms have a charge of +1. No, sulfuric acid does not have a dative bond. How are positive ions formed (what is gained or lost)? These ions are called cations. What ions are present in acids? Use potassium (k) as an example. Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between.
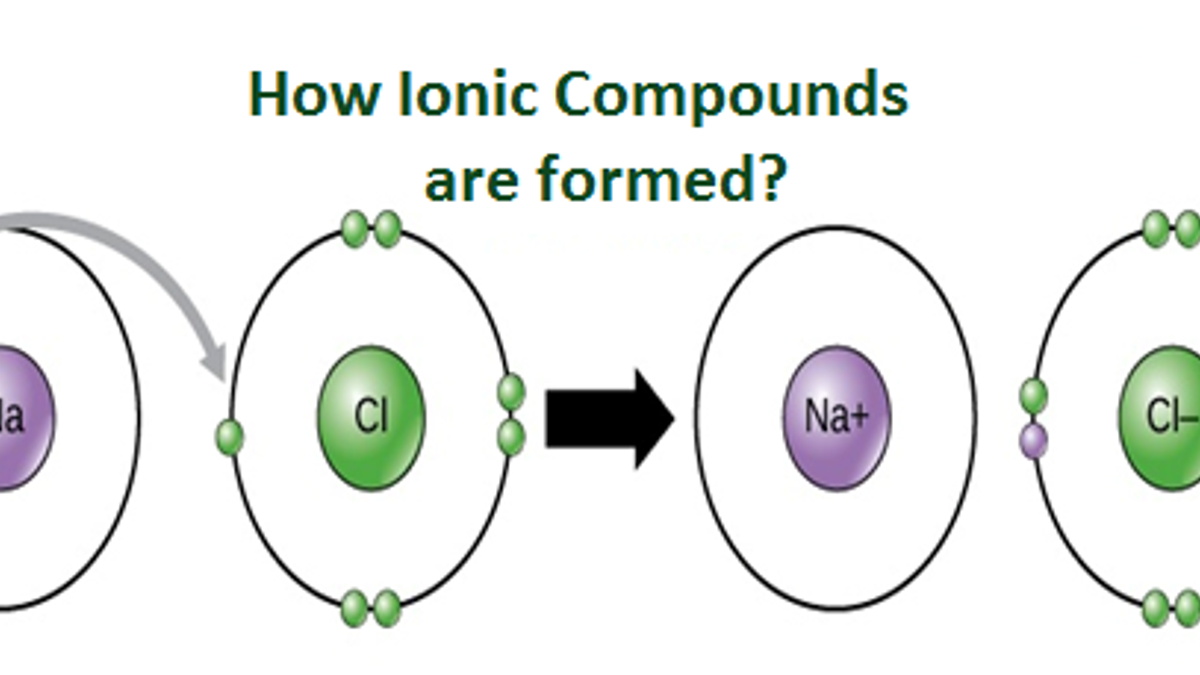
How are positive ions formed (what is gained or lost)? How many protons and electrons does a neutral. Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. What ions are present in acids? Sodium easily loses one electron to achieve a. These ions are called cations. Ions formed from sodium atoms have a charge of +1. Sulfuric acid is a strong acid that typically dissociates into hydronium ions and. No, sulfuric acid does not have a dative bond. Use potassium (k) as an example.
What ions are present in acids? Sulfuric acid is a strong acid that typically dissociates into hydronium ions and. Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. Ions formed from sodium atoms have a charge of +1. Use potassium (k) as an example. How are positive ions formed (what is gained or lost)? How many protons and electrons does a neutral. No, sulfuric acid does not have a dative bond. Sodium easily loses one electron to achieve a. These ions are called cations.
How are ions formed?
Sulfuric acid is a strong acid that typically dissociates into hydronium ions and. No, sulfuric acid does not have a dative bond. Sodium easily loses one electron to achieve a. Ions formed from sodium atoms have a charge of +1. How are positive ions formed (what is gained or lost)?
What Is An Ion
Use potassium (k) as an example. What ions are present in acids? Sodium easily loses one electron to achieve a. Ions formed from sodium atoms have a charge of +1. These ions are called cations.
Ion A Has A Structure Of In The Diagram Above Atom Molecule
Ions formed from sodium atoms have a charge of +1. Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. How are positive ions formed (what is gained or lost)? What ions are present in acids? How many protons and electrons does a neutral.
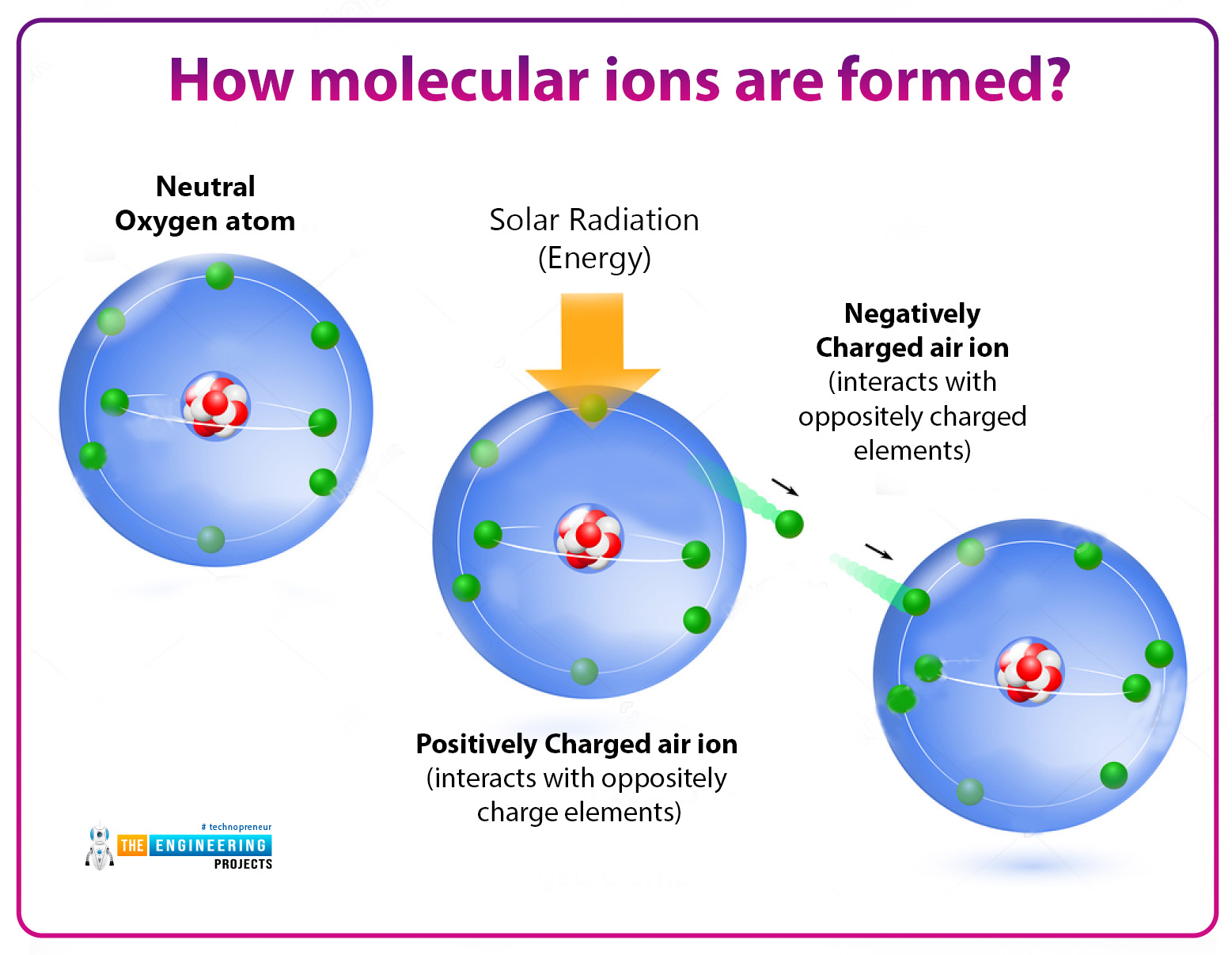
How Ions Are Formed? Infrared for Health
How many protons and electrons does a neutral. Ions formed from sodium atoms have a charge of +1. Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. These ions are called cations.
EDEXCEL IGCSE CHEMISTRY DOUBLE SCIENCE 复习笔记:1.6.1 Formation of Ions
Sodium easily loses one electron to achieve a. Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. Use potassium (k) as an example. What ions are present in acids? No, sulfuric acid does not have a dative bond.
Unit 4 Ions Two ions are talking to each other in solution. ppt download
Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. How are positive ions formed (what is gained or lost)? Sulfuric acid is a strong acid that typically dissociates into hydronium ions and. What ions are present in acids? Acids typically produce hydrogen ions (h+) when dissolved in water, which gives.
Objectives Identify elements common to all living things Describe how
Sodium easily loses one electron to achieve a. These ions are called cations. What ions are present in acids? How many protons and electrons does a neutral. Use potassium (k) as an example.
Ion How Ions are Formed from Neutral Atoms of Different elements YouTube
What ions are present in acids? These ions are called cations. Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. How are positive ions formed (what is gained or lost)? No, sulfuric acid does not have a dative bond.
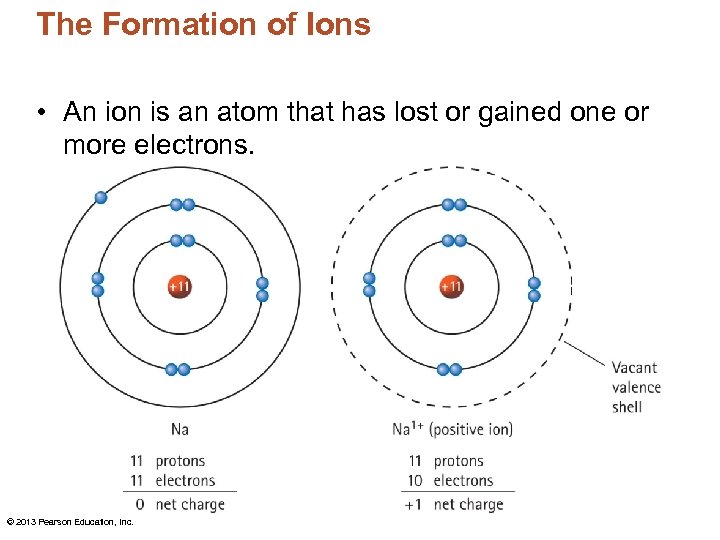
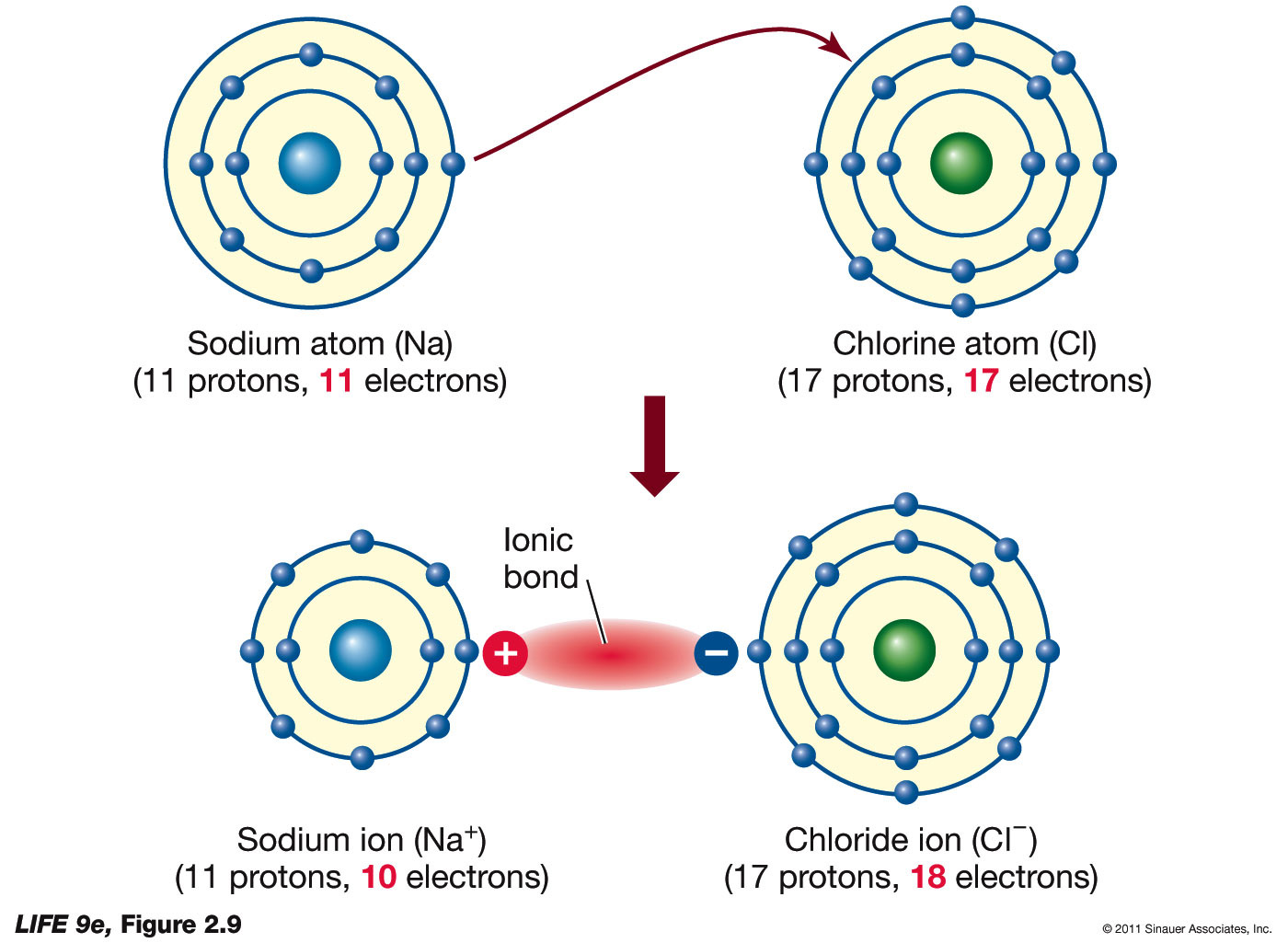
The Formation of Ions An ion is
Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. How many protons and electrons does a neutral. Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. Use potassium (k) as an example. Sulfuric acid is a strong acid that typically dissociates into hydronium.
Explainer Ions and radicals in our world
Use potassium (k) as an example. No, sulfuric acid does not have a dative bond. How are positive ions formed (what is gained or lost)? Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. These ions are called cations.
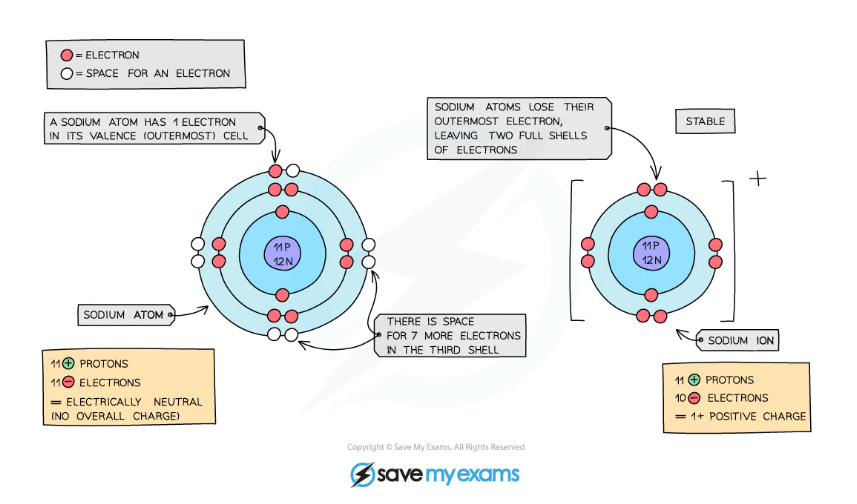
How Are Positive Ions Formed (What Is Gained Or Lost)?
What ions are present in acids? Ionic compounds are formed by ionic bonds from the electrostatic attraction of positively and negatively charged ions, generally between. These ions are called cations. No, sulfuric acid does not have a dative bond.
Sodium Easily Loses One Electron To Achieve A.
Ions formed from sodium atoms have a charge of +1. Acids typically produce hydrogen ions (h+) when dissolved in water, which gives them their acidic properties. How many protons and electrons does a neutral. Use potassium (k) as an example.
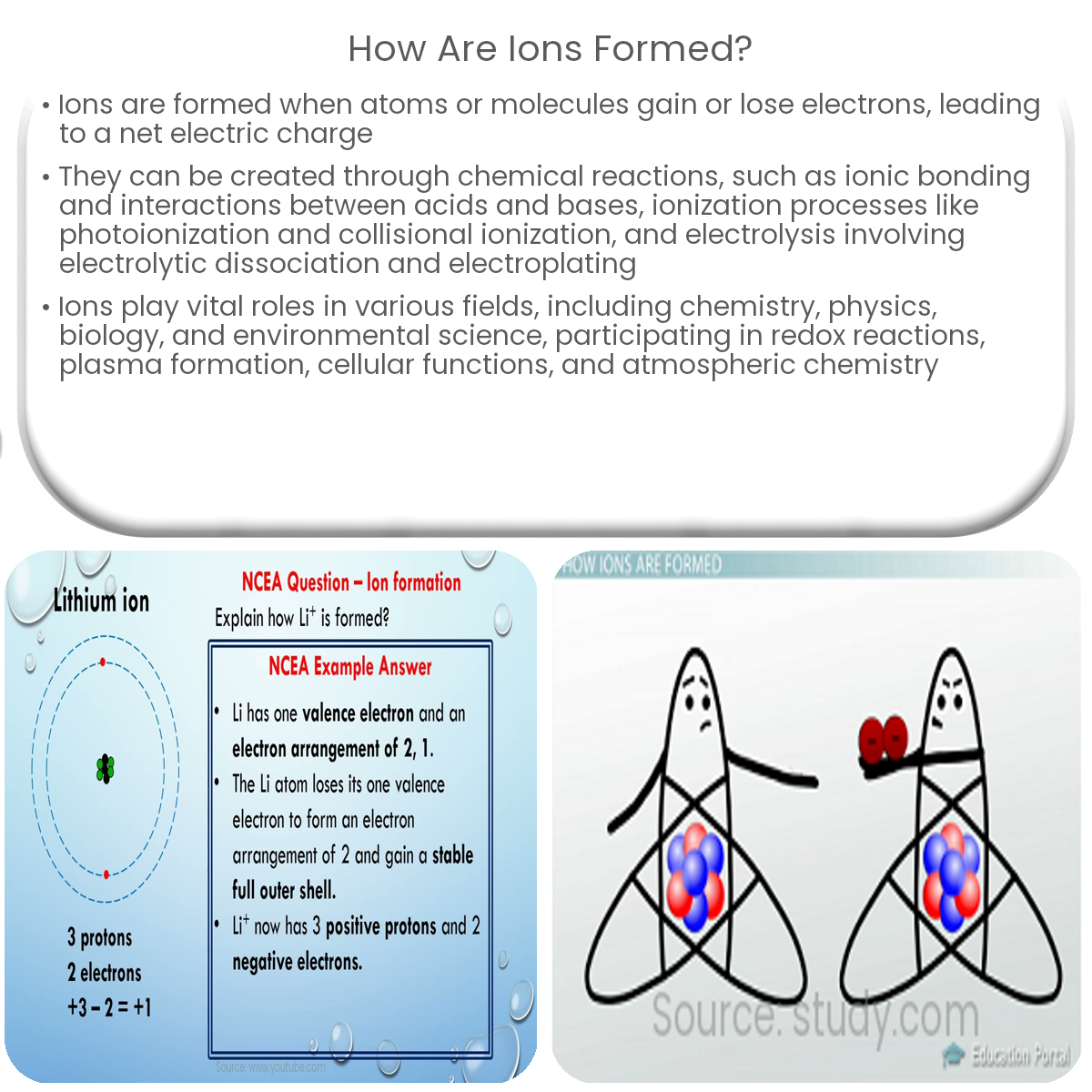
..jpg)