Dimethyl Sulfoxide Safety Data Sheet - How does the reaction proceed?? Is it oxygen lone pair attacks on sulphur which. 1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. I came across the reaction of dimethyl sulfate with water to yield methanol. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. Dimethyl sulphate breaks the oh bond and forms anisole. I understand that the trans isomer doesn't possess.
In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I understand that the trans isomer doesn't possess. Is it oxygen lone pair attacks on sulphur which. I came across the reaction of dimethyl sulfate with water to yield methanol. 1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. How does the reaction proceed?? Dimethyl sulphate breaks the oh bond and forms anisole.
1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. Is it oxygen lone pair attacks on sulphur which. I understand that the trans isomer doesn't possess. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I came across the reaction of dimethyl sulfate with water to yield methanol. How does the reaction proceed?? I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. Dimethyl sulphate breaks the oh bond and forms anisole.
Fillable Online Dimethyl sulfoxide Safety Data Sheet Fax Email Print
How does the reaction proceed?? In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. Dimethyl sulphate breaks the oh bond and forms anisole. Is it oxygen lone pair attacks on sulphur which. I came across the reaction of dimethyl sulfate with water to yield methanol.
DMSO Msds PDF Dimethyl Sulfoxide Toxicity
In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I understand that the trans isomer doesn't possess. Is it oxygen lone pair attacks on sulphur which. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. I came across the reaction of dimethyl sulfate with water to yield.
Dimethyl Disulfide Safety Data Sheet Download Free PDF Toxicity
1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. How does the reaction proceed?? Dimethyl sulphate breaks the oh bond and forms anisole. Is it oxygen lone pair attacks on sulphur which. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells.
Dimethyl Disulfide Safety Data Sheet PDF Dangerous Goods
Dimethyl sulphate breaks the oh bond and forms anisole. Is it oxygen lone pair attacks on sulphur which. 1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. I understand that the trans isomer doesn't possess. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile.
Dimethyl Sulfoxide Baijin
I came across the reaction of dimethyl sulfate with water to yield methanol. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I understand that the trans isomer doesn't possess. Is it oxygen lone pair attacks on sulphur which. How does the reaction proceed??
Dimethyl sulfoxide, properties, uses and safety protection Shenyang
I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. Is it oxygen lone pair attacks on sulphur which. How does the reaction proceed?? 1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse.
(PDF) MATERIAL SAFETY DATA SHEET DMSO … MSDS/DMSO MSDS Liquid
Dimethyl sulphate breaks the oh bond and forms anisole. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. Is it oxygen lone pair attacks on sulphur which. 1 i'm teaching myself organic chemistry and am struggling with differentiating.
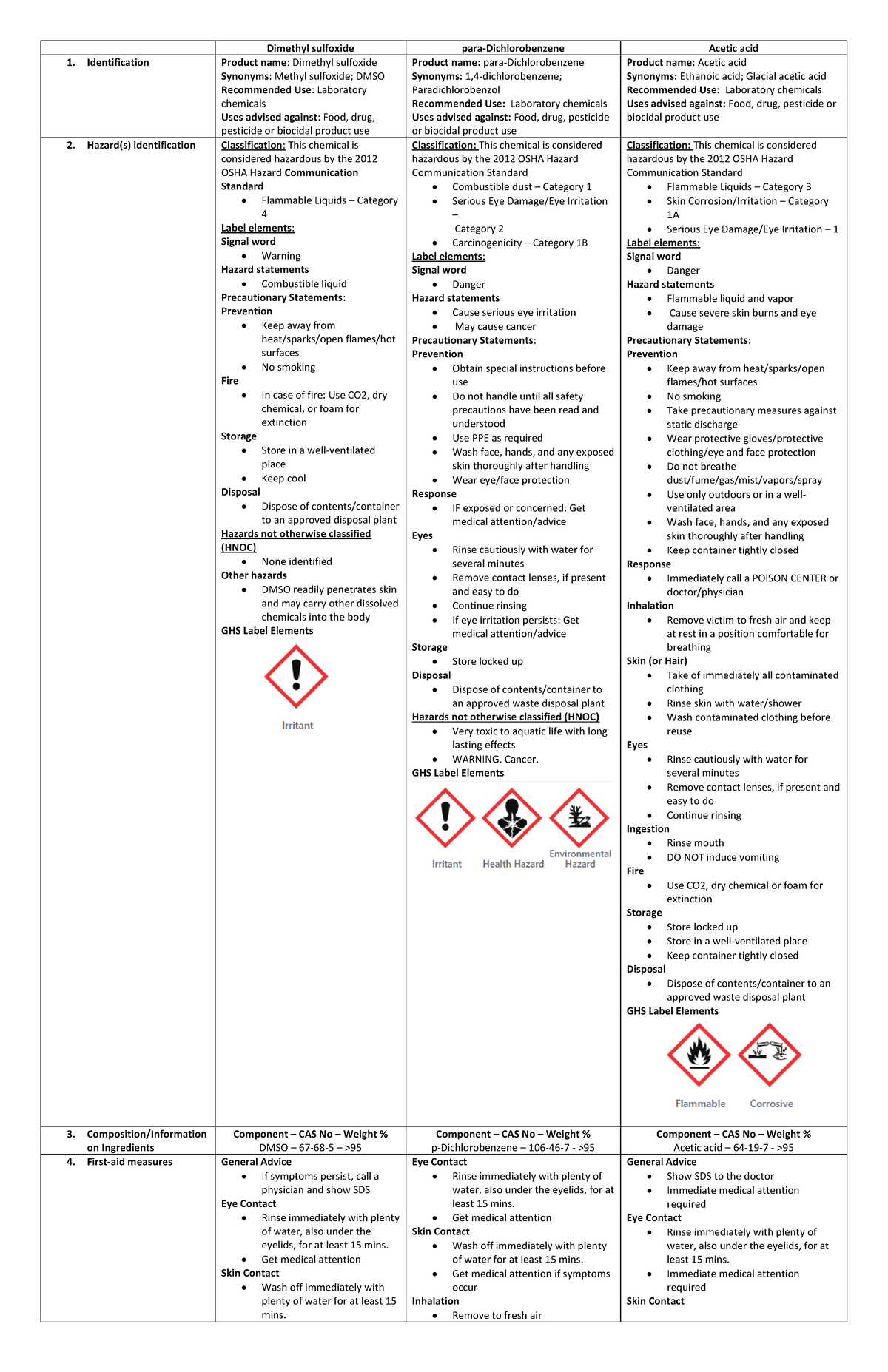
Safety Data Sheets (DMSO, pDichlorobenzene, Acetic acid) Dimethyl
I understand that the trans isomer doesn't possess. Dimethyl sulphate breaks the oh bond and forms anisole. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. I came across the reaction of dimethyl sulfate with water to yield methanol. How does the reaction proceed??
Dimethyl Sulfoxide Baijin
I understand that the trans isomer doesn't possess. I came across the reaction of dimethyl sulfate with water to yield methanol. 1 i'm teaching myself organic chemistry and am struggling with differentiating between dimethyl and ethyl substituent groups. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. Is it oxygen lone pair attacks on sulphur which.
(PDF) Structural parameters of dimethyl sulfoxide, DMSO, at 100 K
I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. Is it oxygen lone pair attacks on sulphur which. I understand that the trans isomer doesn't possess. In cell culture, dimethylsulfoxide (dmso) is added to prevent the formation of ice crystals which may lyse the cells. How does the reaction proceed??
1 I'm Teaching Myself Organic Chemistry And Am Struggling With Differentiating Between Dimethyl And Ethyl Substituent Groups.
Is it oxygen lone pair attacks on sulphur which. I was curious how that works as $\\ce{h2o}$ isn't a strong nucleophile. I understand that the trans isomer doesn't possess. How does the reaction proceed??
In Cell Culture, Dimethylsulfoxide (Dmso) Is Added To Prevent The Formation Of Ice Crystals Which May Lyse The Cells.
Dimethyl sulphate breaks the oh bond and forms anisole. I came across the reaction of dimethyl sulfate with water to yield methanol.







.png)
