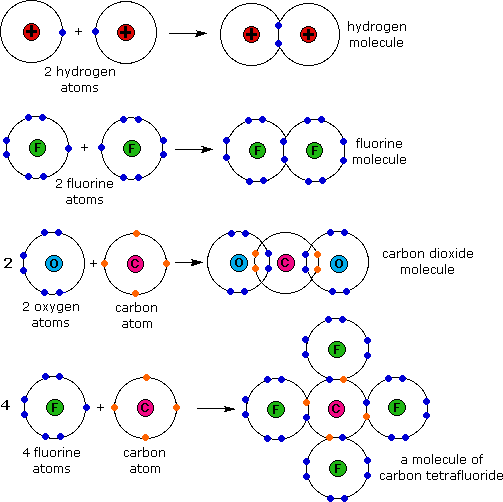
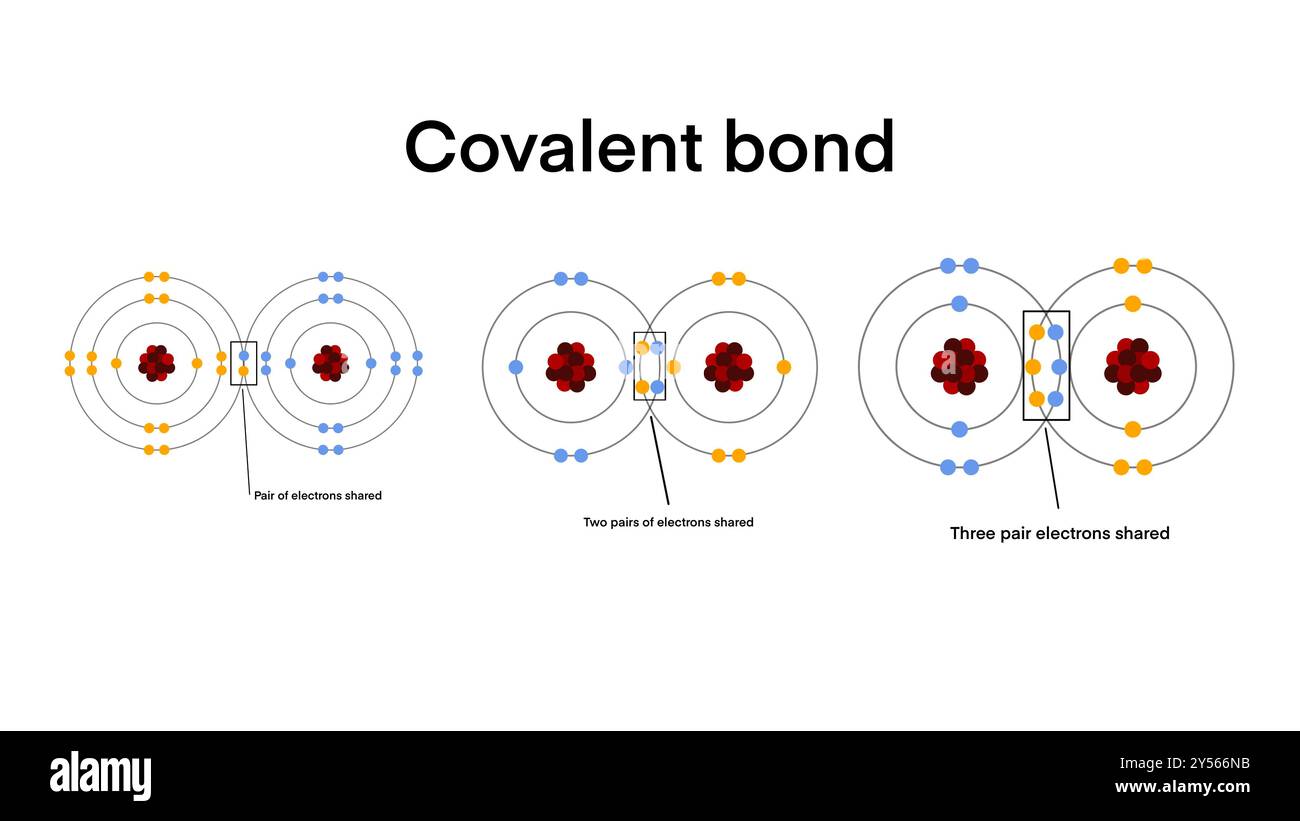
Bond Formed When Electrons Are Shared Between Atoms - A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. What is heterolytic bond breaking? 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. In this bond, both shared electrons are donated by the same atom. It is defined as breaking of.
1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. It is defined as breaking of. What is heterolytic bond breaking? In this bond, both shared electrons are donated by the same atom. A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing.
It is defined as breaking of. What is heterolytic bond breaking? The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. In this bond, both shared electrons are donated by the same atom. 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing.
Biochemistry. ppt download
The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. What is heterolytic bond breaking? 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. It is defined as breaking of. A covalent bond is formed by mutual sharing of two.
The Chemistry of Biology ppt download
A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. It is defined as breaking of. 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. What is heterolytic bond breaking? Heterolytic bond breaking is also known as.
GOOD MORNING!! Lets do a QW!! ppt download
A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. It is defined as breaking of. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5.
What are Chemical bonds? Chemical bonds are formed between atoms when
It is defined as breaking of. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. What is heterolytic bond breaking? 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. Heterolytic bond breaking is also known as heterolysis or heterolytic.
Chapter 2 Chemistry of Life ppt download
1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic..
Electron Configurations & The Periodic Table
What is heterolytic bond breaking? A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. It is defined as breaking of. In this bond, both shared electrons are donated by the same atom.
Covalent Bonding (Biology) — Definition & Role Expii
1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. What is heterolytic bond breaking? A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for.
Chemistry Review ppt download
Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6. It is defined as breaking of. In this bond, both shared electrons are donated by the same atom. A covalent bond is formed by mutual.
covalent bond is a chemical bond that involves the sharing of electrons
In this bond, both shared electrons are donated by the same atom. It is defined as breaking of. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. 1 chemical foundations.
Chapter 2 Atoms and Bonding ppt download
It is defined as breaking of. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. What is heterolytic bond breaking? Heterolytic bond breaking is also known as heterolysis or heterolytic fission or ionic fission. A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving.
Heterolytic Bond Breaking Is Also Known As Heterolysis Or Heterolytic Fission Or Ionic Fission.
A covalent bond is formed by mutual sharing of two electrons between the atoms, each atom giving one electron for sharing. The bond formed between carbon and nitrogen has to be classified as nonpolar covalent, polar covalent or ionic. In this bond, both shared electrons are donated by the same atom. 1 chemical foundations 2 atoms, molecules, and ions 3 stoichiometry 4 types of chemical reactions and solution stoichiometry 5 gases 6.
What Is Heterolytic Bond Breaking?
It is defined as breaking of.