1572 Template - Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. This page provides links to commonly used clinical trial forms relevant to clinical trials. Find the current form, instructions, and. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. For other fda forms, visit the fda forms page. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. Save or instantly send your ready documents. Easily fill out pdf blank, edit, and sign them.
This page provides links to commonly used clinical trial forms relevant to clinical trials. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. Save or instantly send your ready documents. Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. Find the current form, instructions, and. Easily fill out pdf blank, edit, and sign them. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. For other fda forms, visit the fda forms page.
(select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. Find the current form, instructions, and. Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. This page provides links to commonly used clinical trial forms relevant to clinical trials. Easily fill out pdf blank, edit, and sign them. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. For other fda forms, visit the fda forms page. Save or instantly send your ready documents.
Fda 1572 Form ≡ Fill Out Printable PDF Forms Online
This page provides links to commonly used clinical trial forms relevant to clinical trials. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. Easily fill out pdf blank, edit, and sign them. For other fda forms, visit the fda forms page. (select one of the following.) for phase 1.
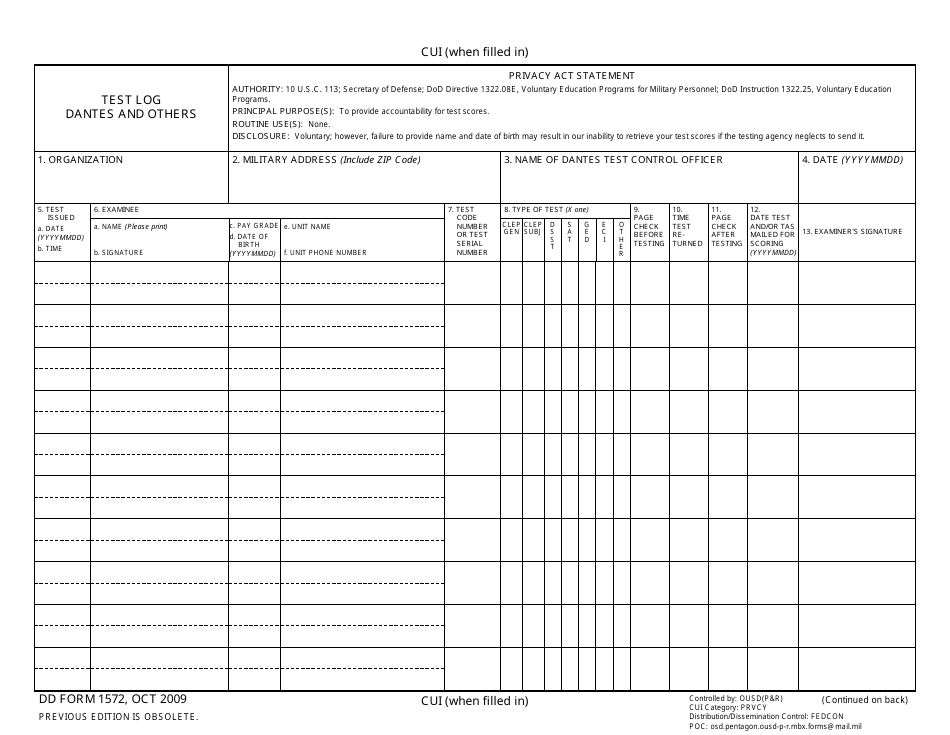
DD Form 1572 Fill Out, Sign Online and Download Fillable PDF
This page provides links to commonly used clinical trial forms relevant to clinical trials. Save or instantly send your ready documents. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. Find the current form, instructions, and. Easily fill out pdf blank, edit, and sign them.
Fda 1572 Template
This page provides links to commonly used clinical trial forms relevant to clinical trials. For other fda forms, visit the fda forms page. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a.
FDA Form 1572 Statement of Investigator Forms Docs 2023
Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. Easily fill out pdf blank, edit, and sign them. For other fda forms, visit the fda.
Fda 1572 Template
Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. Save or instantly send your ready documents. Learn what form fda 1572 is, when and how to complete it,.
Form FDA 1572 Statement of Investigator Free Download
Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. For other fda forms, visit the fda forms page. This page provides links to commonly used clinical trial forms relevant to clinical trials. Learn what form fda 1572 is, when and how to complete it, and what commitments it entails.
Fda 1572 Template
Easily fill out pdf blank, edit, and sign them. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. Save or instantly send your ready documents. Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational..
Top Fda Form 1572 Templates free to download in PDF format
Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. This page provides links to commonly used clinical trial forms relevant to clinical trials. Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. Find the.
Fda 1572 Template
Find the current form, instructions, and. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. This page provides links to commonly used clinical trial forms relevant to clinical.
FDA_1572 Institutional Review Board Health Sciences
Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. Easily fill out pdf blank, edit, and sign them. Find the current form, instructions, and. Save or instantly send your ready.
Find The Current Form, Instructions, And.
Save or instantly send your ready documents. This fda page provides the form fda 1572, statement of investigator, for use in clinical trials and related research. Learn how to complete and sign the statement of investigator, form fda 1572, for clinical trials under a us ind. (select one of the following.) for phase 1 investigations, a general outline of the planned investigation including the estimated duration of.
Easily Fill Out Pdf Blank, Edit, And Sign Them.
Learn what form fda 1572 is, when and how to complete it, and what commitments it entails for investigators of clinical studies of investigational. This page provides links to commonly used clinical trial forms relevant to clinical trials. For other fda forms, visit the fda forms page.